Structure in Chemistry
Macromolecules< >
MM3. Glass Transition
Note: comprehension of this section may be aided by an understanding of conformational analysis.
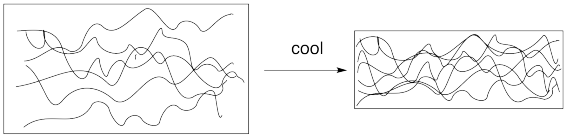
Even though it appears to be a solid, a plastic bottle that you might throw into your recycling bin has some things in common with liquids. We think of the molecules in a solid as mostly static; maybe they rock back and forth a little bit, but they mostly stay put. The long chains in a polymer are constantly in motion, however.
These chains slide over each other like snakes in a process called "reptation" . This flowing motion of the chains is responsible for the flexibility of many plastics, which can often be bent easily even when they are solids.

The ability of polymer chains to move past each other has noticeable effects on polymer properties. When we exert a force upon a polypropylene bottle, it changes shape. Rather than being locked together, the polypropylene chains give way and move past each other. When we let go, the chains may just slide back to their original position, or close to it, returning the bottle to its original shape.
The chains slide back because they were originally in a stable conformation. Rapidly deforming the plastic twists the chains into less favourable positions. Left alone, they twist back into positions that are energetically more favourable.
Of course, it is always possible that the plastic is deformed a little too far and doesn't bounce back to its original shape. In that case, the chains have become stretched out in such a way that they have become stuck. They may have become tangled in new ways. New obstacles have appeared that make it difficult for them to slide back again.
Sometimes, a force is exerted on a plastic material over a long period of time, causing the chains to slowly slide away from their original positions. The chains adopt a new equilibrium distribution. When the force is removed, the plastic has adopted a new shape. A formerly straight bar might become bowed under a weight, for example. This process of a polymer changing shape over time is called "creep".
Problem MM3.1.
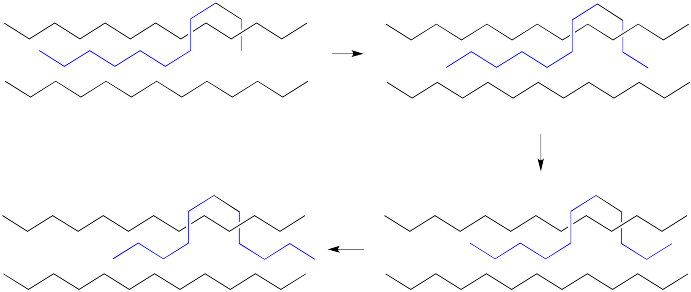
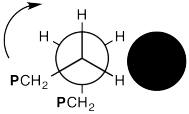
In the drawing below, we are looking down a C-C bond in a polymer chain (left). We can see hydrogens attached to the carbon in front and the carbon behind, although the carbons are not labelled. We can see the where the polymer chain is attached, labelled "CH2P". A cross-section of the neighbouring chain is also shown (in black).
Show, with the help of drawings, how a 360° rotation about that bond will cause the polymer chain on the left to creep over the one on the right.
One of the most commonly-used diagnostic tests for a polymer is the measurement of the glass transition temperature (Tg). The glass transition temperature is related to the ease of chain flow in the material.
The glass transition represents a phase change. As a polymeric material is cooled, it changes from a more flexible, rubbery state to a more rigid, glassy state. This transition occurs because of the change in volume with temperature. Generally, solids shrink as they are cooled. At some point, the volume of a polymer decreases to the point at which the chains can no longer move past each other; there just isn't enough room.
The glass transition temperatures of a number of common polymers are listed below. These data indicate whether a given polymer will exist in a rubbery state or a glassy state at a particular temperature.
| Name | Abbreviation | Approx. Tg, °C |
| low-density polyethylene | LDPE | -120 |
| polyethylene oxide | PEO | -50 |
| polypropylene | PP | -10 |
| nylon-6 | - | 45 |
| polylactide | PLA | 65 (L isomer) |
| poly(ethylene terephthalate) | PETE | 70 |
| polyvinylchloride | PVC or V | 90 |
| polystyrene | PS or S | 100 |
| poly(methyl methacrylate) | PMMA | 110 |
| polycarbonate | PC | 145 |
| polynorbonene | - | 215 |
| kevlar | - | 240 |
These values are only approximate. The glass transition temperature actually depends on a number of factors, including the average molecular weight of the polymer and how the data is measured.
Problem MM3.2.
Explain why glass transition temperature would depend on molecular weight.
Problem MM3.3.
Glass transition temperature is influenced by several factors, including the following ones.
a) sterics (crowdedness, or physical interference between groups as they move through space)
b) polarity
c) backbone rigidity (ability for bond rotation along backbone)
In each case, explain how the factor would influence the glass transition temperature, and choose some examples from the table to support your claim.
Problem MM3.4.
Polymers also undergo other phase transitions. For example, the melting point of polystyrene is around 240 °C. Explain, with the help of drawings, the difference between the glass transition temperature and the melting temperature.
Problem MM3.5.
Indicate whether the following materials would be found in a rubbery state or a glassy state at a comfortable room temperature, about 25°C.
a) LDPE b) PS c) PVC d) PP
Problem MM3.6.
Indicate whether the following materials would be found in a rubbery state or a glassy state at the boiling point of water, about 100°C.
a) PEO b) PETE c) PMMA d) PC
Problem MM3.7.
Why do you think PVC is used in household plumbing, rather than other materials such as PLA or PEO?
This site is written and maintained by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's University (with contributions from other authors as noted). It is freely available for educational use.

Structure & Reactivity in Organic, Biological and Inorganic Chemistry by Chris Schaller is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported License.
Send corrections to cschaller@csbsju.edu
Navigation: