
Reactivity in Chemistry
Aliphatic Nucleophilic Substitution
NS12. Elimination
Sometimes, elimination reactions occur instead of aliphatic nucleophilic substitutions. In an elimination reaction, instead of connecting to the electrophilic carbon, the nucleophile takes a proton from the next carbon away from it. The halide or other leaving group is still displaced. A double bond forms between the two carbons.

Figure NS12.1. An elimination reaction.
Thus, there are actually more than two competing mechanisms occurring at once here. In addition to unimolecular and bimolecular substitution, a reaction involving deprotonation is also possible.
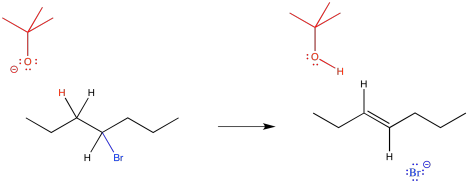
Figure NS12.2. A more detailed look at an elimination reaction.
Instead of acting as a nucleophile, the tert-butoxide anion acts as a base. It forms a bond to a proton, becoming tert-butanol. This proton must always come from the carbon next to the leaving group. The bromide still leaves, and the two adjacent carbons form a second bond together.
Problem NS12.1.
Draw a mechanism for the elimination reaction above. Assume the reaction is bimolecular and concerted, so that the C-H bond and the C-Br bond break at the same time, forming the C=C bond.
The mechanism of an elimination reaction is almost exactly the same as an aliphatic nucleophilic substitution, except that the nucelophile misses its mark. It hits a proton instead of a carbon and acts as a base instead of a nucleophile. This process can happen at the same time as the leaving group's departure or it can happen afterwards. These mechanisms are called E1 and E2.
Problem NS12.2.
Draw another mechanism for the elimination reaction above, but this time, suppose the reaction is unimolecular.
Problem NS12.3.
Given the mechanism in NS12.2., other products would also be expected.
a) What are they? (Think about the reactive intermediate and what else could happen to it.)
b) What does their absence in the original reaction scheme above suggest about the most probable mechanism of the reaction?
Why might a reaction undergo elimination rather than substitution? The most important reason concerns the nature of the nucleophile. The more basic the nucleophile, the more likely it will induce elimination.
What makes something basic, rather than nucleophilic? As a very rough rule of thumb, we can beging by thinking of bases as less stable versions of nucleophiles. Nucleophiles are very often anions, and bases are generally less stable anions. So what factors make anions more stable? Those factors would make the anion more like a nucleophile and less like a base.
Polarisability is one major factor in anion stability. Remember, polarizable atoms are large atoms. In the main group of the periodic table, they include sulfur, phosphorus, chloride, bromide, iodide, etc. These anions are stable because the negative charge is spread out over a larger atom. Any time charge is spread out, it tends to result in greater stability. On the other hand, smaller, less polarisable atoms include oxygen, nitrogen and carbon. Anions of carbon, nitrogen and oxygen tend to be more basic. Anions of bromine, iodine, and sulfur are not basic.
Anions such a bromide and iodide are almost never bases. Smaller, less-polarizable fluoride ions could be basic, but even these ions are more likely to be nucleophiles rather than bases. So if polarizability tells us anything, it is that halides and other stable anions are not very likely to cause eliminations.
You might remember polarizability from a discussion of anion stability in acid-base chemistry. Other considerations from acidity will be useful, too.
Relative electronegativity within a row plays a very significant role in determining whether something is basic. Remember, within a row of the periodic table, size changes are minor. Differences in polarisability are not much of a factor. Instead, differences in electronegativity influence anion stability. The more electronegative the atom, the more stable the anion. As a result, a fluoride anion is more stable than an oxygen anion. Fluoride is more likely to be a nucleophile but we are used to thinking of hydroxide ions as basic. However, an oxygen anion is more stable than a nitrogen anion or carbon anion. As a result, we sometimes make a distinction here, referring to hydroxide ion and alkoxide ions as strong bases, but amide anions and alkyl anions as very strong bases.
Nitrogen anions can be very strong bases. These compounds feature unstabilized nitrogen anions. Examples include amide ion (unfortunately, this is a term from inorganic chemistry that has exactly the same name as an organic functional group that also contains nitrogen). Variations include lithium diisopropylamide (LDA) and potassium hexamethyldisilazide (KHMDS).
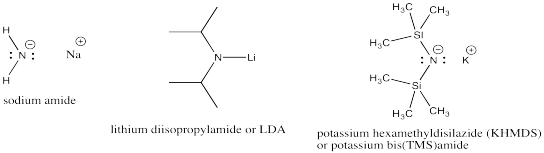
Figure NS12.3. Very strong nitrogen bases.
Very strong bases also include carbon anions or semi-anions. Examples include butyllithium, tert-butyllithium and methyllithium. These compounds are so strongly reactive that they can be very dangerous if not handled correctly. They are all basic enough to react with moisture in the air, deprotonating water molecules and forming hydroxide ion. The reaction produces a flammable hydrocarbon side product and is also very exothermic, so exposure of these compounds to air results in them catching fire. Very strong bases are highly likely to engage in elimination, rather than substitution.
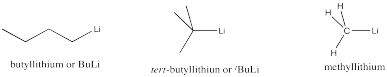
Figure NS12.4. Very strong carbon bases.
Resonance was another important factor that helped to stabilize anions. If a negative charge can be delocalised via resonance, the anion becomes much more stable. For example, an alkoxide ion, such as methoxide, might be very basic, because the negative charge is on an oxygen atom. Oxygen is not a large, polarisable atom. However, an adjacent carbonyl in an acetate anion makes all the difference. This anion is resonance stabilised.
A fourth factor sometimes plays a role in the case of carbon or nitrogen. It is the idea of hybridization. Remember that the geometry of an atom determines which atomic orbitals are involved in bonding. Tetrahedral carbons are thought of as sp3 hybridised, meaning that the carbon uses an s electron and three p electrons in sigma bonding. A trigonal planar carbon is sp2 hybridised. That means it uses only two p orbitals and an s orbital in forming sigma bonds.
Different hybridization leads to some subtle differences in properties. For example, the C-H bonds of sp2 carbons are a little stronger than those of sp3 carbons (maybe 105 to 110 kcal/mol for the former, and 95 to 100 kcal/mol for the latter). That's because the electrons in the sp2 C-H bonds are at slightly lower energy. That, in turn, is because an s orbital is a little lower in energy than a p orbital.
For similar reasons, an sp2 carbanion is more stable than an sp3 carbanion. The electrons on the sp2 carbon are lower in energy than the electrons on the sp3 carbon. An sp carbanion is more stable, still. As a result, although an ethyl anion (CH3CH2-) is extremely basic and a vinyl anion (H2C=CH-) is still highly basic, an acetylide or alkynyl anion (HC=C-), though basic, is much more nucleophilic than the other two.
If any of these three factors apply (anion stability because of polarisability, resonance, sp hybridization in carbon), the anion is often used as a nucleophile rather than a base.
It almost goes without saying that a neutral compound -- in the sense that the compound that has no charge at all -- does not require charge stabilisation. Thus, if a nucleophile has no charge, it is relatively stable, and will often act as a nucleophile rather than a base.
In all of these cases, the nature of the nucleophile is just one factor determining the course of the reaction. Other factors can outweigh the nucleophile, however. As a result, there are examples where these nucleophiles could act as bases. We will see those other factors in a minutes.
Before going on, there is a little more to say about basicity. We talked about some examples of very strong bases already. Let's add a few examples of things that could still be bases but that might not be as strong.
Strong bases include non-stabilized oxygen anions. Examples include sodium hydroxide as well as alkoxides such as potassium tert-butoxide or sodium ethoxide. Strong bases favour elimination, too. Nevertheless, they can sometimes undergo either elimination or substitution, depending on other factors (see below).
Weak bases include cyanide, stabilized oxygen anions such as carboxylates and aryloxides, sulfur anions, fluoride ion and neutral amines. Weak bases are much more likely to undergo substitution than elimination, unless there are additional factors that favour elimination.

Figure NS12.5. Some weak oxygen bases.
So, we have very strong bases, strong bases, and weak bases. We could extend this scale of basicity even further. Very weak bases include heavy halides such as chloride, bromide or iodide, as well as neutral phosphorus or sulfur nucleophiles. Very weak bases undergo elimination only rarely.
Now we can put together an overall summary of what kinds of compounds are likely to undergo eliminations, what kinds of compounds are likely to undergo substitutions, and what kinds of compounds could do either, depending on other factors. Very strong bases such as carbon and nitrogen anions are very likely to undergo elimination. This might not happen if there are additional factors that stabilize the charge, such as resonance delocalization or a high s character in the lone pair (in the case of carbon). Any of the weak bases are more likely to undergo substitution than elimination. Hydroxides and alkoxides are in the middle; they might do either reaction, depending on the conditions.
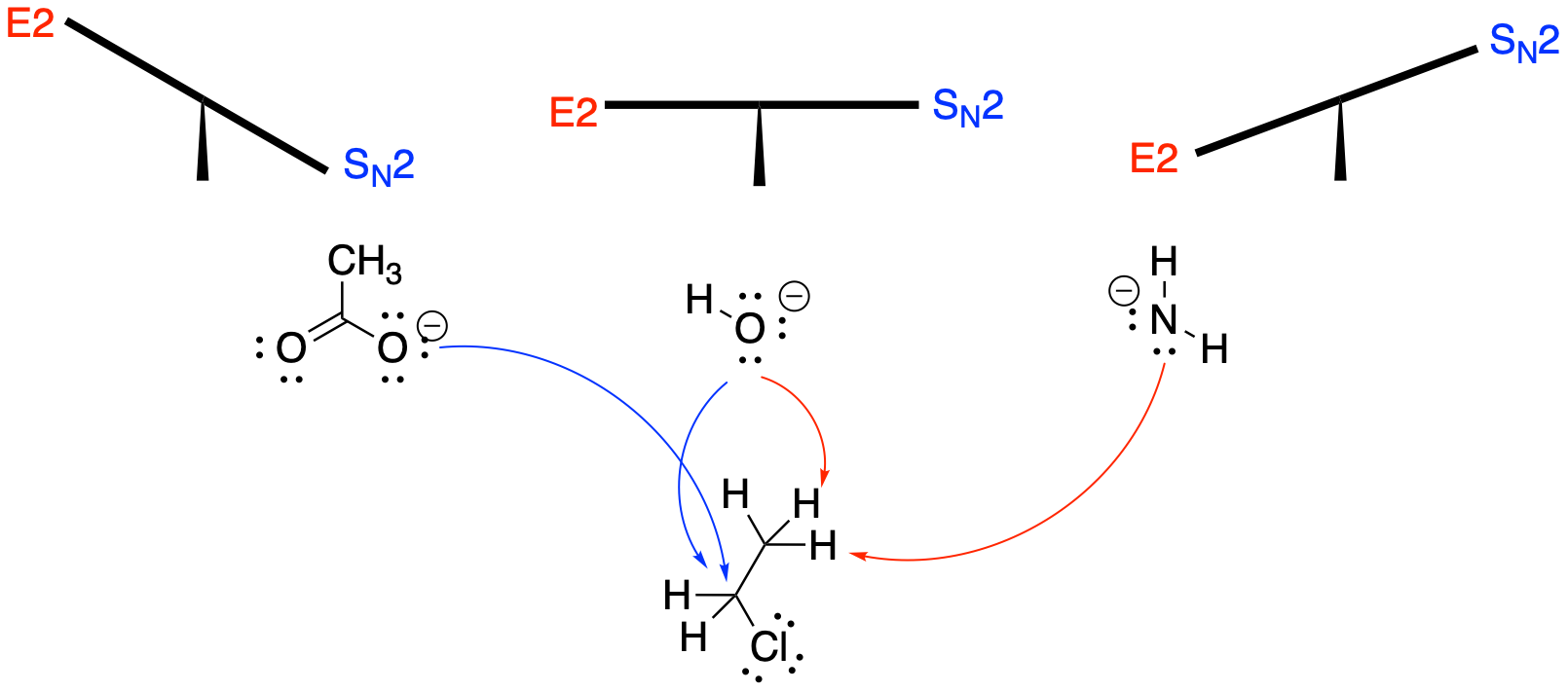
Figure NS12.6. The course of the reaction depends on base strength.
Problem NS12.4.
Classify the following anions as very strong base, strong base, or weak base.
a) NaNH2 b) NaOH c) CH3CO2Na d) NaH e) CH3OH
f) NaBr g) CH3COCH2Li h) CH3CH2CH2CH2Li i) KI j) H2O
k) CH3CCNa l) (CH3CH2)3N m) Na2CO3 n) LiCl o) CH3OK
p) CH3C6H4ONa q) NaSH r) [(CH3)2CH]2NLi s) (CH3)3P t) (CH3)3COK
Problem NS12.5.
Typically, strong bases and very strong bases are more likely to react via the E2 mechanism; they react so quickly that the deprotonation step triggers C-LGp ionization, rather than the other way around. However, E1 mechanisms also occur with strong bases in water if the base concentration is low. Explain why.
Problem NS12.6.
Why is it that an anion such as cyanide is a weak base, whereas CH3Li is a strong base?
Strong bases such as alkoxides will undergo elimination or substitution depending on additional factors. Sometimes, even weak bases can be involved in elimination reactions. Usually, temperature is an important factor in these cases. Fluoride might act as a nucleophile in a reaction with an alkyl halide at 25°C, but as a base in the same reaction at 100°C. Hydroxide or methoxide might behave in exactly the same way: substitution if kept cool, elimination if heated.
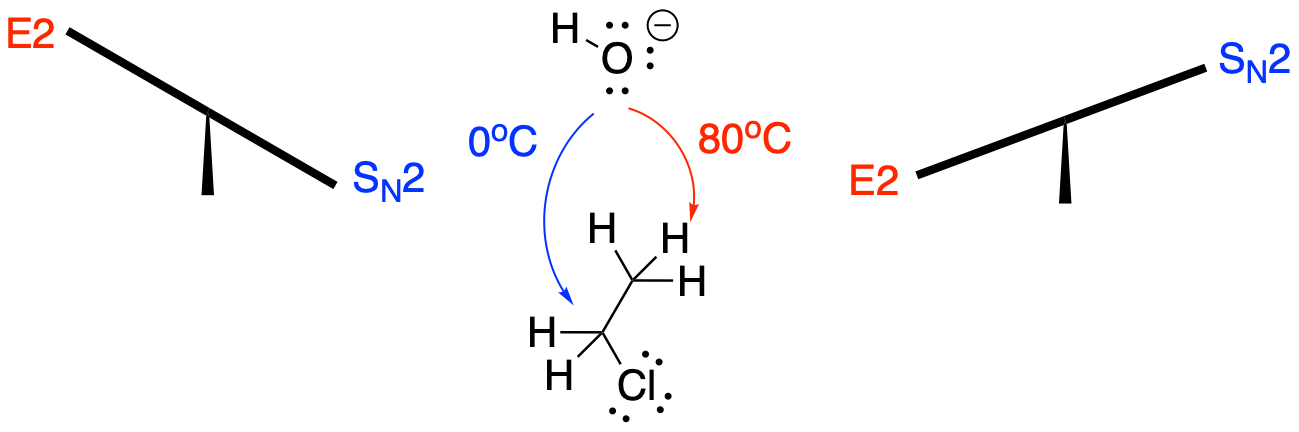
Figure NS12.7. The course of the reaction depends on base strength.
So, why the difference? In part, it has to do with the amount of bond-breaking in the reaction. An elimination reaction involves the cleavage of two bonds, whereas a substitution reaction requires only one bond to break. Thus, an elimination reaction is more energy-intensive, and it is more likely to occur at higher temperatures, when more energy is available. As you will discover below, entropy is an even bigger factor in making elimination more likely at high temperature.
That's not the only reason elimination is favoured at higher temperature. The most important factor in the temperature dependence of eliminations and substitutions is probably . A simple rule we often use to determine whether a reaction is entropically favoured is simply to count the number of species on each side of the equation. Is the number of compounds increasing? Entropy is increasing, and that's favourable. Is the number of compounds decreasing? Entropy is decreasing, and that's unfavourable. Is the number of compounds staying the same? Entropy is not changing very much, and that's neither favourable nor unfavourable.
If you compare an elimination reaction to a substitution reaction, the entropy appears to increase in the elimination reaction, but it doesn't change much in the substitution reaction.
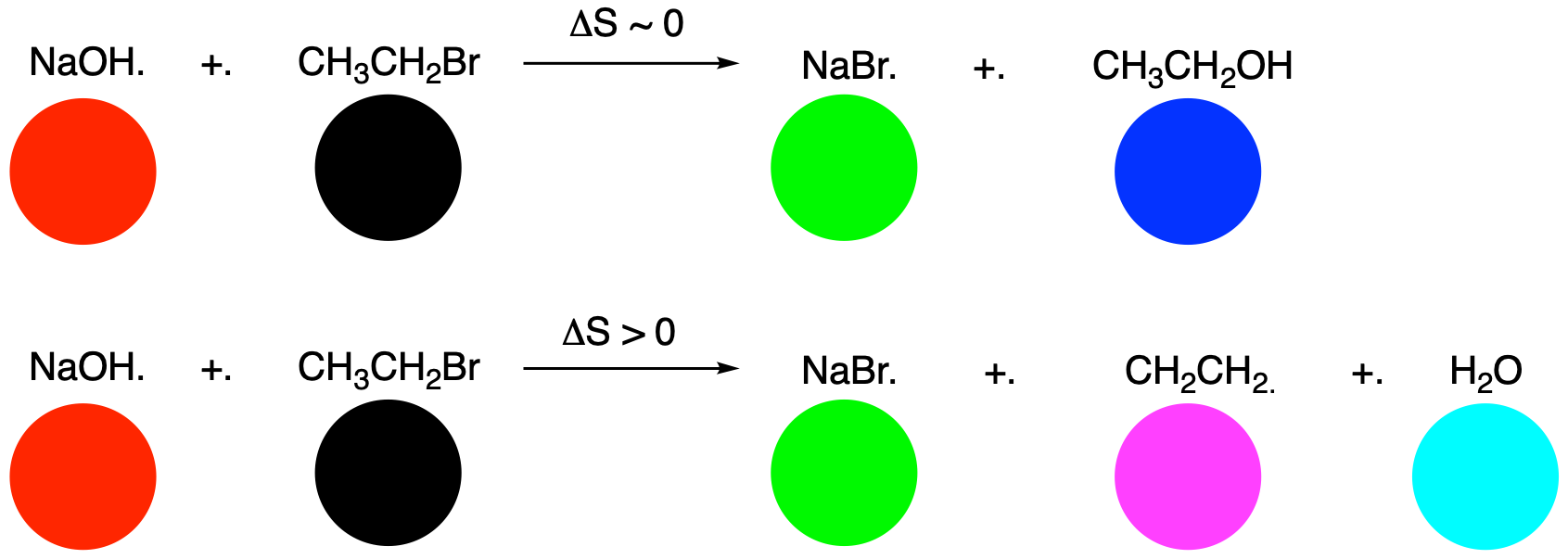
Figure NS12.8. The entropy increases more in an elimination reaction than a substitution reaction.
What does that have to do with temperature? It's all about how free energy works. Given the relationship ΔG = ΔH - T ΔS, which thermodynamic factor dominates free energy change at high temperature? At high temperature, the entropy term, ΔS, becomes much more dominant, because it is being multiplied by the temperature. As temperature increases, entropy gains more weight in influencing the free energy. Therefore, increasing the temperature amplifies the importance of the entropy change in the elimination reaction, so that pathway is more likely.
Steric crowding is another factor that could result in an unexpected elimination reaction. The more crowded the electrophile, the more likely the nucleophile will encounter a proton on its way to the electrophilic carbon.
As a nucleophile approaches tert-butyl bromide, coming from the side opposite the bromine in order to undergo nucleophilic substitution, it is pretty likely to collide with a proton on its way to the electrophilic carbon. The same thing has a good chance of happening with iso-propyl bromide. However, it is much less likely to happen with bromoethane. Finally, bromomethane doesn't even have a beta-hydrogen, so the chance of elimination in that case is zero.
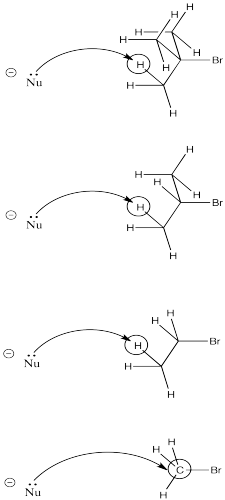
Figure NS12.9. Increased crowding makes it more likely that the nucleophile or base encounters a proton rather than the electrophilic carbon.
Note that the crowding could involve either the structure of the base or the structure of the electrophile. A large, bulky base may be more likely to deprotonate than find its way in to the electrophilic carbon atom.
Problem NS12.7.
Given the following pairs of nucleophiles, which one is more likely to undergo elimination?
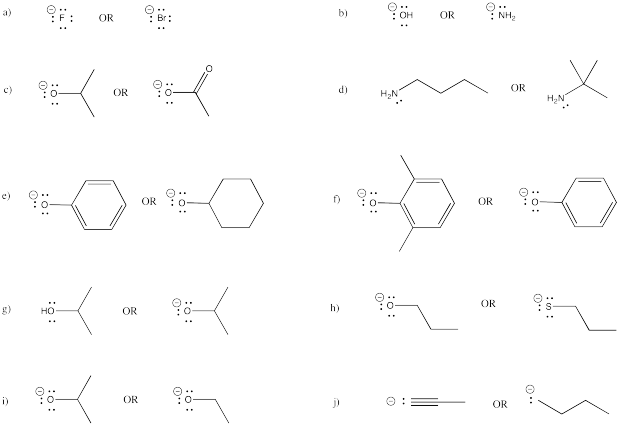
Problem NS12.8.
Although acetylides (such as sodium acetylide, Na CCH) are actually more basic than alkoxides (such as sodium isopropoxide, Na OCH(CH3)2), acetylides frequently undergo substitution rather than elimination. Propose a reason for this difference.
Problem NS12.9.
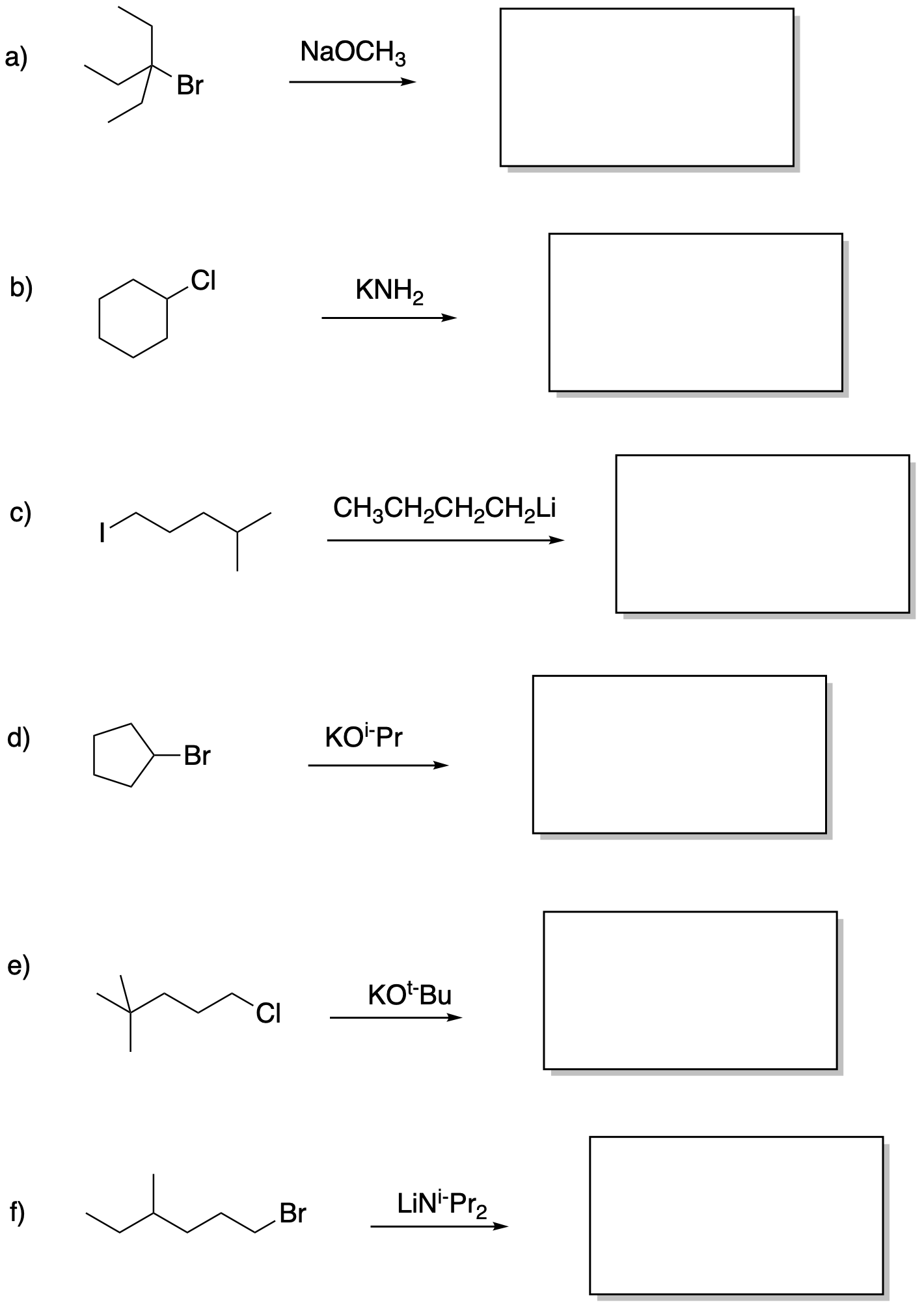
Draw the products of these elimination reactions.
This site was written by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's University (retired) with other authors as noted). It is freely available for educational use.

Structure & Reactivity in Organic, Biological and Inorganic Chemistry by Chris Schaller is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported License.
Send corrections to cschaller@csbsju.edu
This material is based upon work supported by the National Science Foundation under Grant No. 1043566.
Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Navigation: