Structure in Chemistry
Conformational Analysis
CA14. Application Problems
Problem CA14.1.
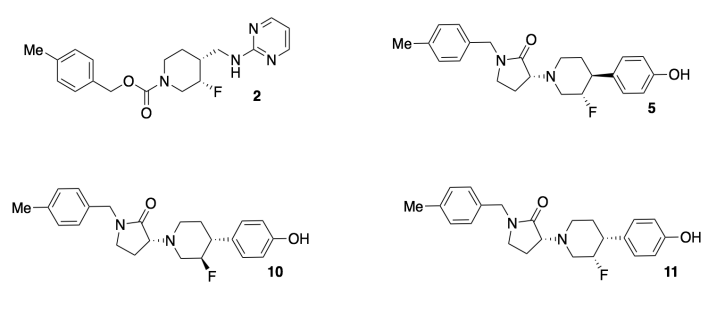
Researchers at Bristol-Myers-Squibb developed a new antidepressant (compound 2, below). The drug binds to GluN2B receptors, shutting down a central nervous system signal that has been linked to depression. Unfortunately, heart problems appeared as a side effect of the medication; the drug also binds to hERG receptors, interfering with potassium ion channels that are important for heart function.

The team designed a new set of compounds (including 5, 10, 11, above. Adapted with permission from Lawrence R. Marcin, Jayakumar Warrier, Srinivasan Thangathirupathy, et al, ACS Med. Chem. Lett. 2018, 9, 472−477. Copyright 2018 American Chemical Society. Used with permission.).
They wanted to make sure the new compounds would still fit tightly in the GluN2B receptor, which is about 9-11 Ångstroms long.
a) On compound 5, circle bonds that run roughly parallel to the length of the molecule (i.e. left-right; don�t circle any that are more up-down).
b) Count up the number of these in a row. If each one is about 1 � long, estimate the length of the molecule.
The functional groups are also important because they control interactions with the receptor.
c) On compound 10, circle and name four of the functional groups. Place an asterisk beside any functional groups found in both compounds 2 and 10.
d) What other factor are the researchers clearly looking at between compounds 5, 10, 11? Why would the researchers be interested in that factor?
e) State the relationships between compounds 5, 10, 11.
f) Find three chiral centers in compound 10. Draw enough of the structure around each chiral center to make it obvious which one you are considering. Rank the priority of the substituents. Assign the configuration (R or S) to each center.
Researchers added an extra ring in the new compounds.
g) Circle the new ring in compound 10 that has no equivalent in compound 2.
h) They incorporated this ring because were interested in adding �conformational rigidity�. To illustrate this idea, draw a Newman projection looking down
i) the middle bond of butane ii) any bond of cyclobutane
Explain why a ring introduces �conformational rigidity� or �conformational restrictions�.
i) Why were the researchers interested in adding conformational rigidity?
j) Only one of the rings in compound 5 can adopt chair conformations (the others are flat). Draw the two possible chairs. Asses steric strains in each conformer. Indicate which conformer is more stable.
Compound 5 was the most promising candidate; it required the lowest dose for GluN2B but the highest dose to bind hERG.
Problem CA14.2.
Tramadol (below) is an opioid analgesic. It is used to treat chronic pain. It is administered as a racemic mixture; each compound targets a different pain receptor.
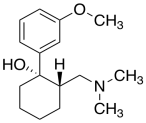
a) Draw the enantiomer and any diastereomer(s) of the molecule shown. Label the configurations at the chiral centers.
b) If the original molecule is part of the therapeutic tramadol mixture, which other compound is also administered?
c) The original compound contains a ring that could adopt different chair conformers. Draw both conformers and assess steric strain in each.
d) Which conformer is the more stable one?
e) The most stable conformer is not necessarily the medicinally active one. How would dosage need to be adjusted if the least stable conformer were actually the active form?
This site was written by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's University (retired) with other authors as noted on individual pages. It is freely available for educational use.
 Structure & Reactivity in Organic, Biological and Inorganic Chemistry
by Chris Schaller is licensed under a
Creative Commons Attribution-NonCommercial 3.0 Unported License.
Structure & Reactivity in Organic, Biological and Inorganic Chemistry
by Chris Schaller is licensed under a
Creative Commons Attribution-NonCommercial 3.0 Unported License.
Send corrections to cschaller@csbsju.edu
Navigation: