Reactivity in Chemistry
Reactions Under Orbital Control
OC1. Introduction
Pericyclic reactions differ from the ones we have looked at so far because they are not easily understood in Lewis acid- Lewis base terms. There is not always a clear nucleophile and electrophile in these reactions. In fact, they may appear to involve completely non-polar reactants.
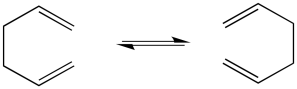
The classic example of a pericyclic reaction is a Cope rearrangement. In a Cope rearrangement, a molecule reorganizes itself, trading in some sigma bonds for pi bonds, so that by the end of the reaction the atoms are connected in a slightly different way. At first, it doesn't seem like much has happened in this reaction, but if you look closely, you can see how specific bonds have changed places.

Figure OC1.1. A very simple Cope rearrangement in 1,5-hexadiene.
Highlighting specific bonds with colour is helpful here. Keeping track of electrons with arrows usually helps, too.
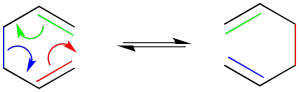
Figure OC1.2. The Cope rearrangement highlighted with colour.
Cycloadditions are very closely related to pericyclic reactions. The most common example of a cycloaddition reaction is a Diels Alder reaction. A Diels Alder reaction takes place between two alkenes. Below, you can see a Diels Alder reaction between ethene and 1,3-butadiene.
Figure OC1.3. Diels Alder reaction between ethene and 1,3-butadiene.
Once again, keeping track of electrons can help us to establish what is going on here.

Figure OC1.4. Diels Alder reaction with electron movement highlighted.
Normally, we think of both of these compounds as nucleophiles. It isn't easy to see why one would react with the other. It isn't easy to see how electrons would be attracted from one molecule to the other.
Instead, pericyclic reactions rely on weak attractions between (or within) molecules that can lead to electronic interactions that result in new bond formation. Normally, pericyclic reactions are studied using molecular orbital calculations to map out these electronic interactions. That analysis makes use of group theory, the mathematics of symmetry. However, they are also explained qualitatively using simplified molecular orbital pictures; that's the approach we'll take here.
Problem OC1.1.
Pericyclic reactions depend on molecular orbital
interactions. Draw the π-bonding molecular orbitals for butadiene, ranked
in energy. In other words, draw a Huckel molecular orbital diagram for
butadiene. Problem OC1.2.
Draw a Huckel molecular orbital diagram for hexatriene. This site is written and maintained by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's
University (retired) with contributions from other authors as noted. It is freely
available for educational use.
This material is based upon work supported by the National Science Foundation
under Grant No. 1043566. Navigation:
Structure & Reactivity in Organic, Biological and Inorganic Chemistry by Chris Schaller is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported License.