02/08/2008
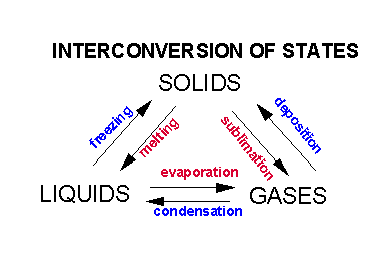
Everyone has learned that there are three states of matter - solids, liquids, and gases. For the rest of the semester we will be discussing small molecules that are held together by covalent bonds, or ionic bonds. Given the property of solids, liquids (take shape container, can be poured, etc) and gases (fill their container), we surmised that the molecules in a solid and liquid must attract each other, with forces that are much weaker than the forces which attract atoms to each other within a molecule - such as covalent bonds. These INTERMOLECULAR attractive forces must be stronger in solids, weaker in liquids, and mostly nonexistent in gases.
Using water as an example, we reviewed how solids could be convert to liquids and then to gases. Indeed, as we saw in the guide on atoms and atomic structure, each state can be interconverted to the others.

It requires energy in the form of heat to change water from a solid to liquid and then to a gas. This energy is required to break up the intermolecular forces which hold the water molecules together. What is the nature of these intermolecular forces?
To answer this question, let's compare the properties of several pairs of molecules. These properties can to a significant degree be determined from the Lewis structures of the molecules.
CO2 and H2O, each with 3 atoms -
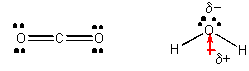
From the Lewis structures we can determine that the geometry of carbon dioxide atoms is linear but the geometry of the water atoms is angular. This leads to the prediction that CO2 is nonpolar but H2O is. It has a permanent dipole. Since all interactions in chemistry are essentially electrostatic in origin, we would hypothesize that IMFs would also arise to some kind of electrostatic interactions. Polar water molecules should attract each other more strongly than the nonpolar carbon dioxide molecules attract each other. This would lead us to further hypothesize that water has a high melting point (MP) and boiling point (BP) than CO2. This is true. Dry ice, which is CO2(s), actually sublimes (turns directly from a solid to a gas) at a temperature much below 0oC., while water melts at 0oC. It takes much less energy to changed states in a substance in which the IMF's are weak than in a substance that can attract other like molecules with stronger IMF's. If water were linear instead of bent, it would have a very low MP and BP and not exist in the liquid state at room temperature, making life on earth impossible.
Let's take a closer look as to how water molecules attract each other. H is the smallest of all atoms. When covalently bonded to O is has a clear δ+ charge. Because it is so small, it can get very close to an oxygen atom on another water molecule. It actually can get very close to a lone pair of electrons on the other O atom. Attractions between + and - and δ+ and δ- depends on how close they get. The closer, the stronger the attractions. Since H is so small and can get so close to a lone pair on an oxygen on another water molecule, the interactions between the δ+ on H and δ- on an O are strong (but much weaker than a covalent bond). This type if intermolecular force is called a hydrogen bond (H-bond).
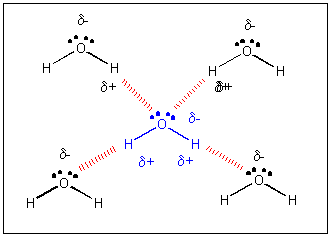
H-bonds can from between an H on a(n) F, O, or N on one molecule, and a partially negative F, O, or N on another molecule. For instance, H-bonds can form between NH3 and H2O, between HF and H2O, but not between F2 and H2O since the F atoms in F2 are not slightly negative or positive since the bond between them is nonpolar covalent. A different way to consider an H bond is that described by Atkins:
"A hydrogen bond is a link formed by a (slightly positive) hydrogen atom lying between two strongly electronegative atoms." (This would include an H bond between the H on water and a Cl- ion, for example.)
H2O and (CH3)2CO (acetone), each a liquid at room temperature -
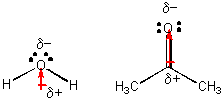
A quick demonstration shows that acetone evaporates much more quickly than water, suggesting that the IMF among acetone molecules are weaker than among water molecules. Both of these molecules are polar, as illustrated above. so what is the difference? Hopefully you can see that water molecules can attract each other through H-bonds, but acetone can't since it has no H's that are bonded to F, O, N, or Cl - i.e. there are no slightly positive H atoms. Acetone molecules attract each other since they are both have permanent dipoles. This type of IMF, which is weaker than H bonds, is called dipole-dipole interactions. You might also be able to imagine an ion - dipole interaction
N2 and NaCl, each with 2 atoms (or ions) -
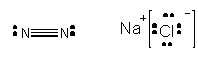
Obviously, N2 exists as a gas at room temperature while NaCl is a solid. Clearly, the IMF's between molecules of NaCl in a crystals of NaCl are much stronger than for N2. Liquid nitrogen exist, but boils at a temperature of -196oC. The difference can be explained by viewing a model of the crystal structure of NaCl.
. Each Na+ was surrounded by 6 Cl- and vice versa. The are very strong IMF's between "molecules" of NaCl in the solid. If you isolate one molecule of NaCl in the crystal structure, it is attracted to other NaCl "molecules" in they solid by ion-ion IMF. This type of IMF clearly is stronger than a H-bond since the attractions are between fully charged ions, not partially charged atoms. In contrast, N2 is not polar and has no permanent dipole. Hence these molecules are attracted to each other weakly. But you know they still attract each other since liquid nitrogen exists. What is the basis for this interaction?
If all attractive interactions arise from charge interactions, then we might speculate that somehow a temporary development of partial charge might develop in nitrogen molecules. You could image this happening in the following ways. Remember, in contrast to our Lewis structures of molecules which show electrons as static bonds or lone pairs, the electrons are actually moving all around the nuclei. They most probably are symmetrically distributed around the molecule. However, at any give time, they would have a probability of being non-symmetrically distributed. For example, at one instance, more of the electrons might be at one end of molecule, giving it a slight negative charge and the opposite end a slight positive charge. That is, a instantaneous dipole is formed. If at that moment, another nitrogen atoms approaches, the slight positive end of the first nitrogen molecule would attract the electron cloud of the second, creating a temporary induced dipole in that molecule, which would allow both molecules to be attracted to each other. This weak IMF is called an induced dipole-induced dipole IMF. It also goes by two other names, Van der Waals forces (VDW) or London Force.
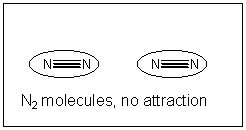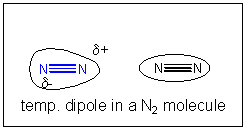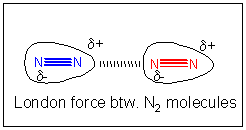
London forces are the only interaction that exist between all species, including ions, polar molecules, and nonpolar molecules. London interactions between polar molecules is usually stronger than their dipole-dipole interactions. This can be seen in the trend in boiling points in HCl, HBr, and HI. Although HCl is more polar than the others, it has a lower BP. HI has the highest BP in this series, because of its large number of electrons, and greater London forces.
The example with acetone above is only partially true. In addition to dipole-dipole interactions, there are more electrons in acetone than water, which would allow greater London forces between acetone molecules than among water molecules. Acetone molecules are attracted by both dipole-dipole interactions and London forces. The strength of the H-bonds among water molecules still predominates in determining the higher boiling point of water compared to acetone. Other types of mixed interactions can also occur.
CH4 and C8H18, each containing just C and H -
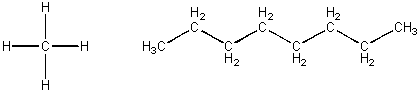
These molecules are both nonpolar and each would attract a like molecule through London forces. The first molecule, methane, is a gas at room temperature. The second, octane, is a liquid at RT and a component of gasoline. Octane molecules must attract each other with strong London forces than do methane molecules. This suggests that the bigger the molecules, the great chance for induced dipoles forming when similar molecules approach. Since all IMF arise from the attraction of + (full, or slight) and - (full or slight), the larger molecule must have more slight + and - interactions another large molecule than occur between two small molecules. With larger molecules, there is greater surface area for these weak attractive forces to work. The table below describes the different types of IMFs and how much energy (kcal is a unit of energy and mol is a a fixed number of such interactions) is required to break the IMFs.
|
IMF |
Energy
(kcal/mol) |
Interacting Species |
|
ion-ion |
60 |
ions only |
|
H-bonds |
4-5 |
FON on 1 molecule and |
|
ion-dipole |
3.5 |
ions and polar molecules |
|
dipole-dipole |
0.5-1 |
polar molecules |
|
London |
0.5 |
all types of molecules |
See the links below for visual applications of IMFs
Quiz: Intermolecular Forces 1
Solutions
Almost all the chemistry that we will study this year deals with reactions of molecules in aqueous solution. This also applies to reactions in the body, which consists of greater than 60% by weight of water. Before we study solutions, we need to review our definition of a solution. In our first unit on matter, we defined solutions as homogeneous mixtures - In homogenous mixtures, the particles are so small that they never separate on standing or in simple centrifugation, and do not interfere with light passing through the mixture. Hence the mixture appears clear. The solution can not be separated into its component parts by filtration. It can be separated by other physical techniques like chromatography (as in lab 1), distillation, etc. If we sample a given solution at different locations, it will have the same composition at every location.
The material that dissolves in a liquid is called the solute. The liquid that dissolves the solute is called the solvent. Of course we can have solution of solids (like salt), liquids (like ethanol) and gases (like carbon dioxide) - all solutes - dissolved in the liquid solvent. Likewise the air is a solution of gas solutes in a gas solvent. Some substance can dissolve in water, others can't. Substances that can't dissolve in water often dissolve in other solvents. Some solid substances dissolve in a solvent like water to different extents. When no more solute can be added to a solvent, we say the solution is saturated with the solute. Any additional solid added will remain as a solid in the solution.
A liquid solute may not dissolve in water. In this case, it will form a discrete layer either above or below the water layer (depending on its density). Such a liquid is immiscible in the solvent. Some liquid solvents dissolve in any proportion in water. Such a liquid solute is completely miscible.
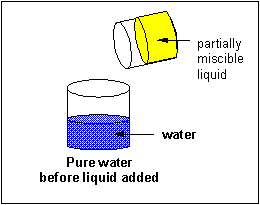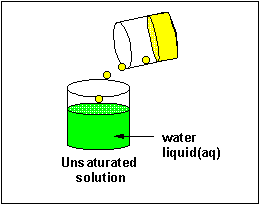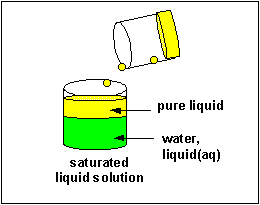
Solubilty of molecules in water and hexane
A knowledge of IMF's can help us understand the solubility of different substances (solutes) in different liquids (solvents). In class I did a series of demonstrations which showed if different solutes were soluble in two different solvents, water and hexane, whose structures are shown below.
A quick inspection of these solvents show them to be quite different. hexane, a clear, colorless liquid at room temperature, is completely nonpolar and interacts with other hexane molecules through London forces. In contrast, water, a clear, colorless liquid at room temperature, is polar and interacts with other water molecules through H-bonds. You might expect they might dissolve different types of solutes. Explore the solubility differences of the different solutes below in hexane and water. Develop a law that will allow you to predict the solubility of a substance in water or hexane, and then develop an hypothesis that helps explain the law.
1. NaCl - sodium chloride:
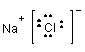
NaCl is a salt held together by ionic bonds. In a sense it is as polar a substance as you can get. It dissolves in water (as you know from experience) but not in hexane.
2. I2 - Iodine:
![]()
Iodine, a covalent solid, in contrast to water, is completely nonpolar. It dissolves in hexane to produce a purple-colored solution. It didn't dissolve in water.
TENTATIVE LAW: From the above two examples, we can surmise that polar molecules dissolve in polar liquids, and non-polar molecules in nonpolar liquids.
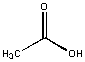
Acetic acid was soluble in water and insoluble in hexane. An inspection of the molecule shows that is is predominately polar with a small nonpolar CH3 group. Our tentative law seems to work so far!
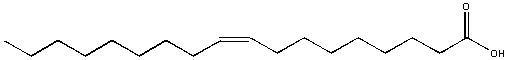
Oleic acid, found in olive oil, is soluble in hexane but not soluble in water. It formed a separate layers in water. In this example, most of the molecule is nonpolar (the long C-H tail) with just a small part at the right end being polar. The nonpolar sections wins out and determines its solubility in water.
MODIFIED TENTATIVE LAW: From the above examples, we can surmise that molecules dissolve in polar liquids if the solute is predominately polar, or in nonpolar solvents if the solute is predominately nonpolar.
![]()
Methanol, a clear, colorless liquid, dissolved in water but only slightly dissolved in hexane. Again this molecule is predominately polar so our modified law is supported. Please note that methanol dissolves but does not form ions in solution.
![]()
Octanol, a mostly nonpolar molecule, dissolves in hexane but formed a separate layer in water. Again our modified law is upheld.
We can use our tentative law to predict whether the following substances are soluble in water or hexane.
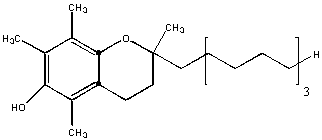
This molecule is almost entirely nonpolar. Hence it will be insoluble in water and soluble in hexane. The is an example of a fat-soluble vitamin. One early definition of fat is biological molecules that are soluble in organic solvents like hexane.
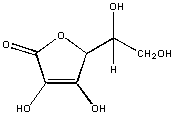
In vitamin C, every C is attached to an oxygen atoms. This molecule is polar and will dissolve in water, but not hexane.
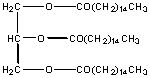
This molecule is also a fat and is the predominant type of fat stored in fat tissue in your body. There are 9 polar carbons, but 42 non polar C's. It is insoluble in water and soluble in hexane. Triglycerides are the major type of substance in vegetable oils.
We arrive at the following conclusions:
REMEMBER, JUST BECAUSE A SOLUTE DISSOLVES IN WATER DOESN'T IMPLY THAT IT DISSOCIATES INTO IONS LIKE SOLUBLE SALTS AND STRONG ACIDS!
Image: How methanol, CH3OH dissolves in water.
You should now be able to predict the solubility of different substances in water and other solvents. Take for instance O2. Would you expect this to be very soluble in water? It's not since it is nonpolar and can't form ion-ion, H-bonds, or dipole-dipole interactions with water. We can't get by by "breathing water" since not enough O2 can be dissolved in water. That's why we have a protein in our blood called hemoglobin which specifically binds and carries O2, effectively increasing the "dissolved" concentration of oxygen by 100 fold. Given this info, how can you explain the photo below:
Quiz: Intermolecular Forces 2 and Solutions
Micelles and Bilayers
We have seen that the solubility properties of molecules strongly depend on how much of the molecule is polar and how much is nonpolar. Consider stearic acid, shown below, which has 18 carbons. The end containing the two oxygens (shown in red) is polar, but the rest of the molecule is completely nonpolar (shown in blue). We can draw a "cartoon" model of this model as a circle - representing the polar end or "head group" with a single connecting line - representing the long, nonpolar "tail".
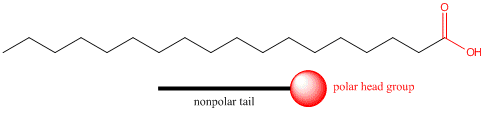
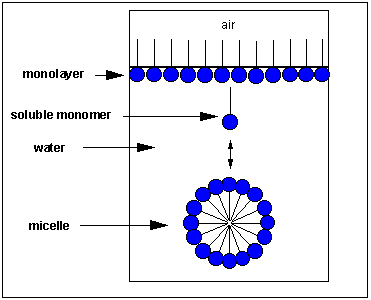
Pretend you are that molecule with your head representing the polar head group and the rest of your body the nonpolar tail. Now dive into water. How would you orient yourself in water? There are several possible ways. A small number of these molecules might be soluble in water (remember even insoluble salts dissociate to a small degree to form a few ions). Mostly the nonpolar tail wants to get out of the water, while the polar head like to stay in the water. There are two ways this can be done. Some of the molecules migrate to the surface of the water, with the nonpolar tails sticking out into air, away from water, to form a monolayer on the top of the water. Others will self-aggregate, through IMF's to form a spherical structure in which the nonpolar tails are sequested from water and the polar head are facing the water. This structure is called a micelle. Detergents consist of molecules with very similar structure to stearic acid pictured above. They form micelles in water. Grease from clothes or foods, normally not soluble in water, can "dive" into the middle of the micelle and be carried off by this structure. Notice that there is not water inside the micelle as you can see by selecting Micelles below.
Now consider another molecule somewhat similar to stearic acid, called a phoshpolipid. A typical phospholipid structure is shown below. Notice it has a polar end (shown in red), but in contrast to stearic acid above, it has two long nonpolar tails shown in blue. We can draw a "cartoon" model of this as a circle - representing the polar end or "head group" with two connecting lines - representing the two long nonpolar "tails".
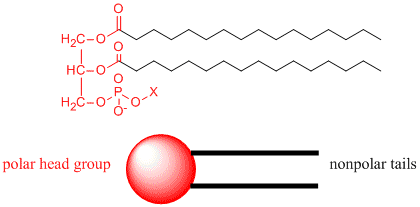
How would you orient this molecule in water? There are several possible ways. A small number of these molecules might be soluble in water as above. Mostly, however, the nonpolar tails wants to get out of the water, while the polar head like to stay in the water. Again, some of the molecules migrate to the surface of the water, with the nonpolar tails sticking out into air, away from water, to form a monolayer on the top of the water. Others will self- aggregate, through IMF's to form a bilayer or membrane. Because there are two tails per head group, the tails can't pack together as tightly. Imagine the bilayer or membrane curving around and eventually meeting. A structure like this would look like a small biological cell. In contrast in a micelle, the interior of this little cell, or liposome, is filled with water which can interact through IMF's to the head groups of the inner leaflet of this membrane. The head groups of the outer leaflet of the membrane interact through IMF's with the bulk water.
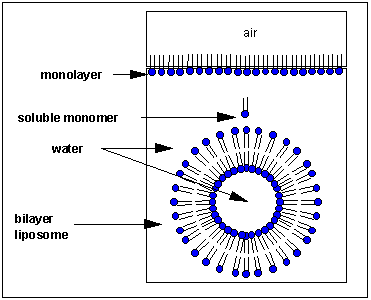
Phospholipids are the basic component of actually biological membranes. Liposomes are useful since they are simple models of actual biological membranes. In addition, they are starting to be used therapeutically. Toxic drugs, like those used in chemotherapy, can be incorporated into the aqueous volume inside a liposome where they can be targeted to specific tumor cells.
Liposomes and micelles seem to be complicated structures. However their formation and existence can be predicted from the simple solubility properties of these molecules and the concepts of IMF's.