Biochemistry Online: An Approach Based on Chemical Logic

CHAPTER 9 - METABOLIC AND SIGNAL TRANSDUCTION
A. ACTIVE TRANSPORT
BIOCHEMISTRY - DR. JAKUBOWSKI
04/16/16
|
Learning Goals/Objectives for Chapter 9A:
|
A1. Introduction to Active Transport
We have previously discussed how chemical potential energy in the form of reduced organic molecules can be transduced into the chemical potential energy of ATP. This ATP can be used to drive reductive biosynthesis and movement (from individual cells to whole organisms). ATP has two other significant uses in the cell.
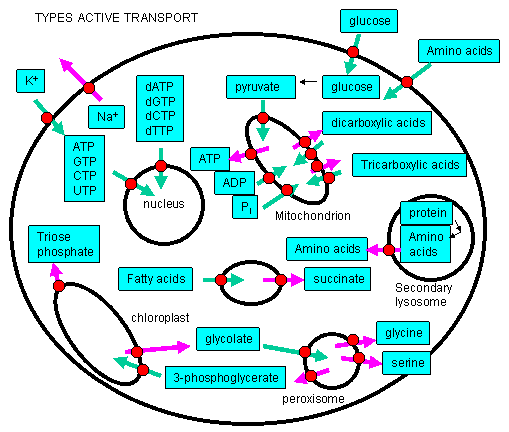
Active Transport: Molecules must often move across membranes against a concentration gradient - from low to high chemical potential - in a process characterized by a positive ΔG. As protons could be "pumped" across the inner mitochondrial membrane against a concentration gradient, powered by the ΔG associated with electron transport (passing electrons from NADH to dioxygen), other species can cross membranes against a concentration gradient - a process called active transport - if coupled to ATP hydrolysis or the collapse of another gradient. This active transport is differentiated from facilitated diffusion we studied earlier, which occurred down a concentration gradient across the membrane. Many such species must be transported into the cell or into intracellular organelles against a concentration gradient!
Signal Transduction: All cells must know how to respond to their environment. They must be able to divide, grow, secrete, synthesize, degrade, differentiate, cease growth, and even die when the appropriate signal is given. This signal invariably is a molecule which binds to a receptor, typically on the cell surface. (Exceptions include light transduction in retinal cells when the signal is a photon, and lipophilic hormones which pass through the membrane.) Binding is followed by shape changes in transmembrane protein receptors which effectively transmits the signal into the cytoplasm. We will discuss two main types of signal transduction pathways:
- nerve conduction, in which a presynaptic neuron releases a neurotransmitter causing a postsynaptic neuron to "fire";
- signaling at the cell surface which leads to activation of kinases within the cytoplasm;
We will discuss signal transduction in the final two chapters.
For active transport to occur, a membrane receptor is required which recognizes the ligand to be transported. Of major interest to us, however, is the energy source used to drive the transport against a concentration gradient. The biological world has adapted to use almost any source of energy available.
Energy released by oxidation: We have already encountered the active transport of protons driven by oxidative processes. In electron transport in respiring mitochondria, NADH is oxidized as it passes electrons to a series of mobile electron carriers (ubiquione, cytochrome C, and eventually dioxygen) using Complex 1, 3 and 4 in the inner membrane of the mitochondria. Somehow the energy lost in this thermodynamically favored process was coupled to conformational changes in the complex which caused protons to be ejected from the matrix into the inner membrane space. One can imagine a series of conformation-sensitive pKa changes in various side chains in the complexes which lead in concert to the vectorially discharge of protons.
ATP hydrolysis: One would expect that this ubiquitous carrier of free energy would by used to drive active transport. In fact, this is one of the predominant roles of ATP in the biological world. 70% of all ATP turnover in the brain is used for the creation and maintenance of a Na and K ion gradient across nerve cell membranes using the membrane protein Na+/K+ ATPase.
Light: Photosynthetic bacteria have a membrane protein called bacteriorhodopsin which contains retinal, a conjugated polyene derived from beta-carotene. It is analogous to the visual pigment protein rhodopsin in retinal cells. Absorption of light by the retinal induces a conformation changes in the retinal and protein, which leads to vectorial discharge of protons ;
Collapse of an ion gradient: The favorable collapse of an ion gradient can be used to drive the transport of a different species against a concentration gradient. We have already observed that collapse of a proton gradient across the inner mitochondria membrane (through FoF1ATPase) can drive the thermodynamically unfavored synthesis of ATP. Collapse of a proton gradient provides a proton-motive force which can drive the active transport of sugars. Likewise a sodium-motive force can drive active transport of metal ions. Since the energy to make the initial ion gradients usually comes from ATP hydrolysis, ATP indirectly powers the transport of the other species against a gradient.
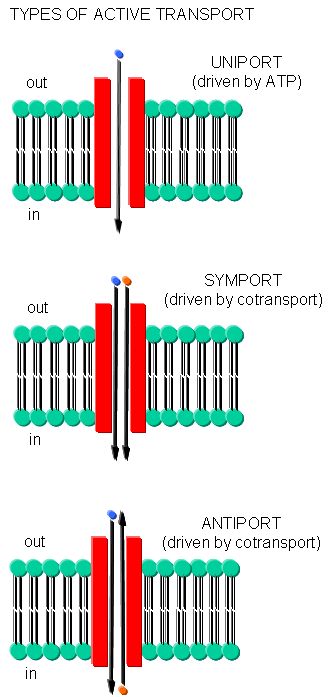
Often times, transport of one species is coupled to transport of another. If the species are charged, a net change in charge across the membrane may occur. Several terms are used to describe various types of transport:
- symport - two species are cotransported in the same direction by the same transport protein
- antiport - two species are cotransported in opposite directions by the same transport protein
- electrogenic - a net electrical imbalance is generated across the membrane by symport or antiport of charged species
- electroneutral - no net electrical imbalance is generated across the membrane by symport or antiport of charged species
Navigation
Return to Chapter 9A: Active Transport Sections
Return to Biochemistry Online Table of Contents
Archived version of full Chapter 9A: Active Transport

Biochemistry Online by Henry Jakubowski is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.