SC13. Macromolecular structures: alpha-helices
Amino acids are very important building blocks. They form larger structures such as polypeptides, oligopeptides and proteins. The first two cases are just terms for chains of amino acids bonded together; the third refers to extremely long chains, and sometimes several long chains wrapped together.
Peptides are formed via amide bonds between an amino group on one amino acid and a carboxylic acid group on another.
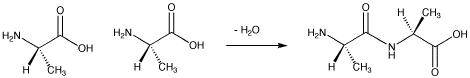
Figure SC13.1. Formation of the peptide Ala-Ala.
Problem SC13.1 Sometimes more than one combination of amino acids is possible in a peptide.
a) Show the two possible combinations of alanine and glycine.
b) Would these compounds have identical properties? Why or why not?
Problem SC13.2. Peptide names are written by describing the chain of amino acids from the amine end of the peptide to the carboxylic acid end. Draw the peptide Ala-Phe-Lys.
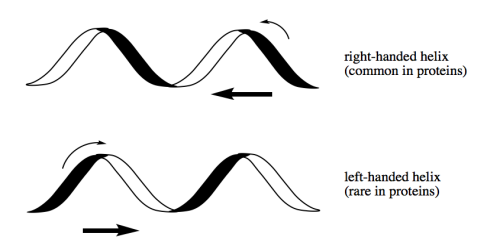
There are a number of possible "macromolecular structures" that peptides and proteins can adopt. One of them is an alpha helix, in which a long chain of peptides coils up like a phone cord (go to a museum if you don't know what a phone cord looks like).
- Alpha helices are a common structural motif in proteins.
- Alpha helices are chiral; a mirror-image form is possible.
- The shape of the alpha-helix is rooted in the shape of the L-amino acids that compose the protein.
The determination of helical structures in proteins was a very recent discovery at the time that Rosalind Franklin obtained x-ray crystallography data on a DNA sample. It was during a visit to her lab that Francis Crick and James Watson began to think that DNA also had a helical structure. Franklin's unusual data resembled results for proteins that Watson and Crick had recently been shown by Linus Pauling. Watson and Crick correctly surmised that DNA had a similar structure. Pauling won the Nobel Prize, twice, Watson and Crick won the Nobel Prize for their work on DNA structure, but Rosalind Franklin unfortunately died very young, and the Nobel Prize is not awarded posthumously. Partly as a result, her name is very often not associated with this important work, despite her crucial contribution.
- Alpha helices are also found in DNA.
- The chiral component of DNA is a chiral carbohydrate.
