Biochemistry Online: An Approach Based on Chemical Logic

CHAPTER 1 - LIPID STRUCTURE
E: Why do Single Chain Amphiphiles form Micelles not Bilayers, and Double Chain Amphiphiles form Bilayers, not Micelles
BIOCHEMISTRY - DR. JAKUBOWSKI
01/25/2012
|
Learning Goals/Objectives for Chapter 1E: After class and this reading, students will be able to
|
Making Liposomes
Micelles and liposomes form spontaneously - i.e. delta G < 0. When water is added to dried phospholipids, multi-lamellar vesicles (MLVs) form, with water trapped between the successive polar head groups of the multiple bilayers. Sonicating these will produce small unilamellar vesicles (SUVs). If however, an aqueous detergent solution (such as beta-D-octylglucopyranoside) is added to a concentration greater than its CMC, mixed micelles of the phospholipid and detergent result. By slowly dialyzing away the detergent, which is selected on the basis of its high CMC, the phospholipids in the micelle aggregate to form a large unilamellar vesicle (LUV). Various drugs may be encapsulated in the liposome by placing them in solution before the dialysis step.
Why do single chain amphiphiles form micelles and double chain amphphiles form bilayers?
As the number of C in the alkyl chain increase, the Δμ for HC transferring into a micelle, or by analogy, for a single chain amphiphile entering a micelle, becomes more and more negative. The following equation seems to apply to the transfer of a single chain amphiphile into a micelle:
Δμo = μo (mic) - μo (aq) = + number - 709 NC
where NC is the number of carbon atoms in the chain. The first positive term depends on the nature of the head group, while the second negative term is independent of the head group. These + and - terms brings us back to the principle of opposing forces we discussed when we looked at the intemolecular forces involved in micelle and bilayer formation. There are attractive IMFs, including van der Waals interactions among the chains and dipole-ion, and H-bond interactions with water and the head groups. There are likewise repulsive interactions arising from steric hindrance with bulky heady groups and ion-ion repulsions. Of course there are also entropic considerations. Let us now consider these factors as we explore what might happen to a preformed micelle as we try to put more single chain amphiphiles (sca) into it. Clearly, as we increase the number of Cs in the sca, the micelles would have a larger radius. For a given sca of fixed number of Cs, once a spherical micelle is formed, it can no longer retain its spherical shape if more sca's are added. Imagine increasing the diameter of a spherical micelle 10x. A large part of the the inside would have no atoms or be filled with water, which would not be favorable. Therefore, if the micelle is to grow, it can do so only by changing shape to something other than a sphere. By squeezing a ball, one can imagine the the shape could distort to a circular cylinder. In this way the acyl chains can still interact. The only problem is that head groups will now be closer than they were in the sphere. Imagine in a sphere the head groups radiating in a perpendicular direction from the sphere surface. As the sphere is distorted to a cylinder, the head groups would come closer together, and hence they will experience more steric interference. If a cylinder can be formed, however, it could continue to grow as long as needed with no further compression required. Imagine now that you further compressed the cylinder into a planar "bilayer" structure. The head groups would be even closer and experience even more repulsion. This bilayer will not form since growth can occur in the cylindrical phase without the added repulsion.
Now consider a double chain amphiphile (dca). In the case of a sca, the number (N) of head groups (HG) = the number of acyl chains (CH). Hence the surface area per HG is equal to the area per HC. Or: As/N HG = As/ N CH. For a dca, N HG = N CH/2, therefore As/N HG = 2As/NCH. There is twice the surface area available per head group compared to that of the sca. Therefore the dca can tolerate more compression. In fact, it can easily be compressed to a bilayer, which as we saw, has much less As/HG. The cylindrical form actually has to much space per head group since water can enter the structure. The extra closeness of head groups in the bilayer can be tolerated even more, since the Dmo for transfer of a dca into a micelle is 60% more negative than that of a sca. The As/HG for closed vesicles differs only slightly from that of a truly planar bilayer since the vesicles are so large compared to a micelle.
Once again, we have discovered that structure mediates function. We can account for the fact that sca and dca form micelles and bilayers, respectively, by understanding the structure of the monomers!
Figure: Surface Area per Head Group vs no. of C in Amphiphile - Globular, Cylindrical, Planar Forms.

In reality, things are more complicated
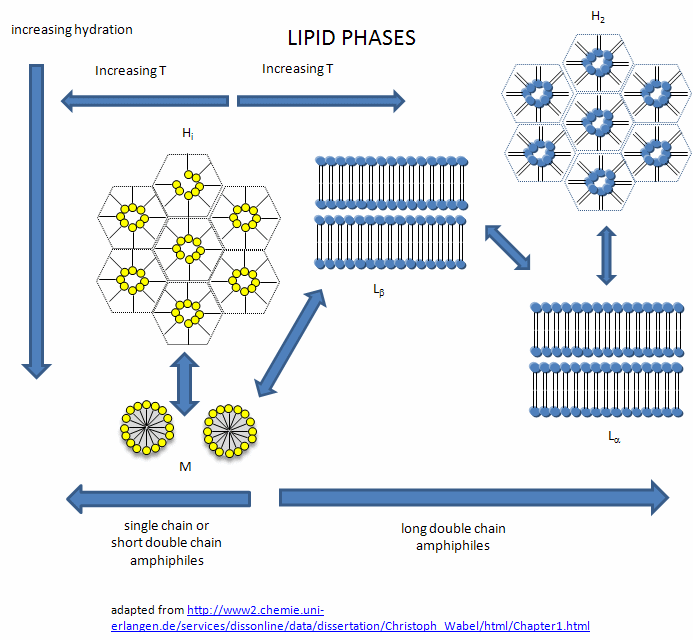
In this introductory chapter on lipid aggregates, I have simplified the nature of the complex lipid structures than can form on aggregation of single and double chain amphiphiles. The general rule holds that single chain amphiphiles form micelles and double chain amphiphiles form bilayers. However, under the right condition, single chain fatty acids can from bilayers, which makes fatty acids like oleic acid prime candidates for components of the membranes of protocells in the evolution of life from abiotic conditions. In addition, other lipids phases can be observed. What aggregates or phase ultimately forms depends on the structure of the lipid, the solvent conditions, and the temperature. These include the following phases:
-
lamellar gel (Lb) and lamellar liquid crystalline (La) phases
-
hexagonal HI (cylinders packed in the shape of a hexagon with polar heads facing out into water
-
hexagonal HII (cylinders packed in the shape of a hexagon with acyl chains pointing out as in reverse micelles, and
-
micellar (M).
Figure: Lipid Phases
.

Biochemistry Online by Henry Jakubowski is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.