HISTORY OF MEDICINE- WEST
Ancient Greek medicine can be divided into 5 stages: Homeric,
Mythological, Pre-Hippocratic, Hippocratic, and Post Hippocratic
HOMERIC MEDICINE: 9th century BC; description based on oral
tradition, archeology, writings.
- Gods cause disease. Iliad opens with epidemic sent by Apollo, the
arbiter of health and disease. Asclepius, his son, was a legendary physician. Hygieria,
his daughter, was goddess of health.
- stop disease by discovering what offends the gods.
- knowledge of anatomy from butchering, sacrifices, and battle injuries
- thymos - life force in all parts of a living organism,
maintained by factors from the outside (food, drink, air) and inside (blood, etc.); could
escape from wounds and by exhaling; leaves body at death
- psyche: soul or individual personality; spirit, which went to
underworld after death; heart source of consciousness
MYTHOLOGICAL:
- Asclepios, god of healing (Apollo's son); a warrior king;
- many temples build to Asclepios, where healing occurred. Patients
leave offering to Gods.
- Many types of illnesses treated, many of which were psychosocial
PREHIPPOCRATIC : 6th century BC
- philosopher/scientists of time used new model: give all phenomena
natural vs. supernatural explanations.
- Thales (around 600 BC) ; thought there was one element, water,
from which came earth, air.
- Pythagoras (530 BC). developed school of philosophy but also
religious cult; Humans were fallen gods, which eventually return back to the divine
through the rebirth of the soul in animals. (Hindu, Buddhist ideal) It was science of
numbers not elements that determined fate. 1 = God, 2 = matter, etc. Seek balance.
Opposite pairs of substance and qualities achieve balance. 4 elements, 4 qualities, hence
4 becomes important in health. Stressed vegetarian diet; some foods used to treat disease.
- Alcmaeon - health is harmony, disease disturbance of harmony.
But need dissection, not just philosophy to understand body. Through dissections (animals)
concluded brain seat of mind. Thought sleep induced when brain blood vessels filled with
blood. Eye contained both fire and water. First real medical scientist.
- Empedocles: (450
BC, see History of Science - West) described 4 elements with 4 qualities, which had a great
effect on the development of medical theory.
SUMMARY OF MEDICAL PRACTICE AT TIME OF HIPPOCRATES:
No longer believed illness caused by Gods, but little effective
treatment available. If patient couldn't be improved or cured, abandoned by practitioners.
Patient in better state since now would be the subject of rational treatment.
THEORY
- men study philosophy, rhetoric, science, and medicine, including
surgery.
- Illness explained in terms of 4 humors (derived logically from
the 4 elements, 4 qualities, pairs of opposites (especially #4) needed to be balanced to
achieve harmony in universe and body; 4 seasons, which affect mind and body; 4 visible
secretions (blood, phlegm, bile - yellow and black)
- all body fluids composed of various proportions of blood (warm &
wet), phlegm (cold and wet), yellow bile (hot and dry) and black bile (cold and dry).
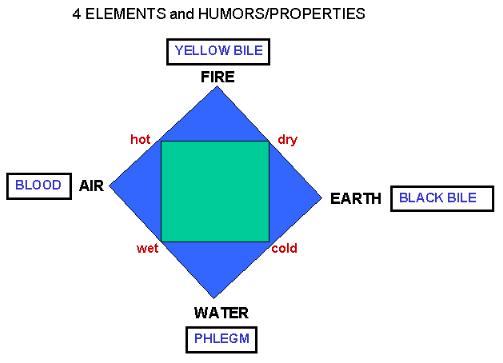
- humors in balance = health; Excess or lack = disease.
- 3 stages of disease:
a.) change in levels of humors caused by external or internal factors;
b.) body react with fever or boiling;
c.) discharge of excess humor which lead to cure, or death.
- often see discharge during illness: blood, phlegm, vomit urine,
sweat, fecal matter
- Causes of mental disease seen less likely as supernatural or demonic,
but more rational; But Plato, contemporary of Hippocrates, used mythology to classify
mental illness in 4 categories: a. prophetic - mediated by Apollo; b. ritualistic ; c.
poetic; d. erotic
TRAINING
- physician status of craftsman from aristocratic class
- poor not likely to afford physician (may have given rise to Asclepian
temples available to all.
- magical medicine available to all
- train by apprenticeship; did house calls since itinerant; operations
often performed in houses
- no system of licensure; anyone except a women could be a physician.
METHODS
- diet, exercise, temperance,;
- numbers important: 4 day interval between fevers; menses - 4X7,
pregnancy - 4x70 days
- wounds cleaned and treated with minerals and plant extracts, which
included wine.
- some medication to induce vomiting; use enemas.
- lots of bone breaks treated with mechanical devises; cauterized
wounds. Gave juice of opium poppy for pain relief.
- physicians care for animals; animal anatomy applied to humans; humans
alone has soul.
- physical exam include questions about life style, emotion state,
behavior, climate;
- successful physician made accurate prognosis, not necessarily cured
patient.
HIPPOCRATES - 460 BC - 370 BC (approx.)

Had most profound influence on Western and some aspects of Eastern
medicine of anyone in history. His esteem increased with time, as his legend (based on
truth or rumor) grew. A collection of his works, "Corpus Hippocraticum" (72
books), probably including contributions from others, was assembled in 4th cent. BC in
library, established by Ptolmey, at Alexandria, Egypt. First to mention 4 humors. Balance of 4 humors determines the state of
the microcosm (body) as the state of the world (macrocosm) was determined by the 4
elements (which were formalized by Aristotle). Ideas in his
books:
- Anatomy: scant information. Heart description quite complete,
including pericardium ventricles, valves, auricles and ventricles. Didn't understand the
differences between veins and arteries.
- Physiology: heat of the body necessary for life comes from the
pneuma of the air, and is taken in by lungs. Air and blood fills arteries. Sight depends
on lens and gel in eyeball. 4 humors corresponding to 4 elements are physiological bases
of body function. Harmony necessary for health.
- Pathology: cause of illness due to internal or external
influences like climate, diet, activity, surroundings. 3 stages of disease: degeneration
of humors; cooking, and crisis with departure of bad humors. 40 case histories.
- Therapy: avoid interfering with course of illness except at
right time. Assist nature in cure. Used few drugs. Laxatives and emetic herbs main agents.
Diet, baths important.
- Diagnosis: list few diseases and syndromes. Condition of
patient most important, so get info. on past and present behavior and problems. Observe
patient and excretions. Orifices probed with speculums. Listen to breathing but not much
info. on heart sounds. Little uses of pulses.
- Prognosis: Use all info. to consider prognosis. Patient
confidence and reputation of physician depend mostly on accurate prognosis.
- Surgery: Most extensive listing. Fractures, dislocations,
wounds of skull given much treatment. Bandaging and cauterization stressed; "What
drugs fail to cure, that the knife cures; what the knife cures not, that the fire cures;
but what fire fails to cure, this must be called incurable. " Treat tumors, ulcers,
hemorrhoids with surgery.
- Gynecology/Obstetrics: fetus thought to initiate
own delivery. Diseases of uterus known but it was thought to wander around (as in the case
of hysteria).
- Mental Illness: Brain was seat of thought and understanding
(later to be changed by Aristotle). Accurate description of epilepsy, deliriums,
depression, anxiety
- Ethics: Exceptional treatment of ethics. Deal with who should
enter profession, how the physician should act, what they should say and do to comfort
patient. Physician should lead a moderate life.
Hippocratic Oath
- describes what a physician should do and not do
- avoid doing harm to patient and life by not giving deadly drugs,
doing abortions or surgery, having sexual relations with patients, discussing patient
information with others.
- duty is to "in purity and holiness I will guard my life and my
art"
- Probably does not originate with Hippocrates since he described
techniques for surgery and abortions
- might have originated with the Pythagoreans who prohibited taking any
life, and who believed the soul was found in blood and hence avoided surgery.
- remained a part of medical doctrine and education possibly through
support o Christian churches.
Hipppocratic Method
Rational methods free of religious or supernatural thinking were
great advance in medical practice.
- OBSERVE ALL: take intensive history. Leave nothing to chance.
Histories are written down in great detail. Physician use all senses in observing patient.
- STUDY THE PATIENT RATHER THAN THE DISEASE: study how the
patient reacted to a disease, not just disease. Surroundings and way of life of patient
very important; Opposed classifying diseases according to organs affected.
- EVALUATE HONESTLY: 40 case histories presented in works, half
of which died. Objective, including lack of response to treatment. Prognosis was most
important, for that is what people really wanted to know.
- ASSIST NATURE: Make conditions good for natural forces in the
body to reach harmony and health. Must leave alone what he cannot help. "As to
diseases, make a habit of two things; to help or at least not to harm".
- weaknesses:
- anatomic knowledge scarce, unsystematic.
- lack of specific diagnosis.
- Let nature take its course led to wait and see mentality.
- supported 4 humor theory
POST HIPPOCRATIC - splits into variety of sects.
Plato - (see History of
Science - West)
- Influenced by Pythagoreans since great interest in numbers; Main
interest in soul and nature of matter; medical thoughts deduced by logic but without
experimental evidence. The Dogmatists in 3rd cent. BC and after used his doctrines
(reasoning superseded observation; experience means of testing, but mostly to support own
ideas) classified all diseases according to humors. Used extreme therapeutic measures -
drastic purging and bleedings
Aristotle (see
History of Science - West);
- wrote extensively on comparative animal anatomy. described beating of
embryo heart, some difference between arteries and veins, the aorta (which he named), that
fetus did not breath.
- But, believed in 4 humors; formalized relationship among 4 humors, 4
elements, and qualities, as pictured above.
- no concept of evolution; imagined all species unchanging
- living things had soul; nonliving hand none; humans most highly
developed soul
- heart seat of intelligence; confused nerves with ligaments and
tendons, linked veins from liver to right arm and from spleen to left arm, which was
theory of bloodletting; dreams portents of the future
- Was an experimentalist, in contrast to Plato, his teacher. Aristotle,
with Hippocrates (past) and Galen (future) were source of most medical authority.
4 Humors in Greater Detail (from
The Greatest Benefit to Mankind - Ray Porter)
Phlegm and bile were associated with disease, since
they became evidence in sickness. Cold in winter were associated with excess phlegm,
while summer diahrrea and vomiting were associated with bile. Of the 60 books in the
Hippocratic Corpus, one, Airs, Waters, and Places details information on disturbance in
the humors. Since blood was released during menstruation, it was likely that blood
letting would also have therapeutic effects. Black bile was seen in feces and vomit.
The four humors could be correlated through analogy to the four qualities, four
seasons, four ages of people (infancy, youth, adulthood, and old age) and four temperments
(melancholyi, choleric, sanguine and phlematic). For example
- yellow bile (hot and dry)
predominated in summer. Severe fevers likely (malaria).
- black bile (dry and cold) increased
in fall as the weather cooled. Fevers waned but after effects (of
malaria) - enlarged spleen (source of black bile), discolored and darkened skin and
darkened urine appeared
- phlegm (cold and moist) predominated
in winter since it was cold and wet, like the Mediterranean climate, so
colds, pneumonia more likely;
- blood (hot and moist) predominated
in spring (and among young); an excess of blood under these
conditions could be avoided by avoiding food rich in blood or by blood-letting; spring
fever (probably malaria), nosebleeds, dysentery more likely.
SUMMARY OF MEDICAL PRACTICE IN ROMAN EMPIRE:
Greek medicine reached peak in Alexandria, and then moved to Rome
which controlled much of the area. Early Roman medicine based on Gods, but eventually gave
way to more rational approaches. Greek culture become predominant part of intellectual
life in Rome. Many Greek physicians came to Rome.
Asclepiades (around 100 BC) -
- repudiated Hippocrates
- believed physician cured disease, not nature
- abandoned 4 humors, and instead devised a "solid" system in
which assume body composed of an almost infinite number of atoms of different size and in
motion (based on Democritus). Between atoms flowed body fluids
- Health depend on smooth activity of atoms; sickness when atoms
disordered.
- Used diet, exercise, massage, medications, enemas, music, and singing
- developed tracheostomy (opening in the wind pipe)
- for "phrentis" (mental illness) used opium and wine
- Overthrow of acceptance of authority, reject humors, use material
approach to body mechanisms major step to rational health care system.
- Galen (below) criticized him for beliefs, and his views dismissed
until the Renaissance.
GALEN - 129-200 (approx.)

Greek physician and most influential writer of all time on medicine.
This work was basis for medicine for 1500 years. Careful and accurate, but uncritical
observations. Worked in Alexandria and Rome. Wrote huge amount about anatomy, physiology,
pharmacology, pathology, therapy, dietetics, hygiene, philosophy, all in Greek, the
language of science. Scholars accepted his work blindly. Considered first
experimentalists.
CHARACTERISTICS OF WRITING
- teleological explanation for everything. He believed everything was
predetermined,which caused him to see fit everything to his views. For example, organs had
function since Nature gave them purpose. These ideas were attractive to Christian church.
Aristotle said "Nature does nothing without a purpose." Galen supplied the
purpose. Galen expanded Aristotelian ideas into medicine. Every structure in a body has
purpose. All things are determined by a wise and perfect God.
- His theory of illness based on Aristotelian 4 humors and previous
Hipppocratic corpus. Used the humor theory of the old Greeks that 4 humors were
responsible for health and disease. He elaborated on it, and proposed there were four
personalities: phlegmatic, sanguine, choleric, and melancholic. He believed fevers resulted from an excess of the biles,
phlegm, or blood, since they excess was stored in certain places of the body where it
would putrify, causing fever. Bloodletting became popular. Mental illness was
also characterized. Mania (a hot illness caused by yellow bile and treated with
cooling agents) and Melancholia (depression - caused by black bile) were common labels
used for mental illness. .
- concentrated on anatomical detail. Saw veins connected to heart, and
nerves coming from central nervous system. Showed the anatomy of the spinal cord.
Dissected accident victims and abandoned corpses, but based much anatomy on animals.
- Although much data accurate, made many mistakes in internal organs.
Would propose structures not there to fit theories such as holed between left and right
sides of heart.
- Discerned importance of emotions and symptoms and believed in
prevention through hygiene.
- His physiology based on the idea that the "life principle"
was a spirit drawn into the body from the spirit world while breathing.
To describe the body, Greeks and Romans used
metaphors derived form domestic scenes and agriculture. Galen thought the slow flow of
blood to nourish the limbs and organs was like a great irrigation system. He thought the
blood didn't circulate. Originates in liver, goes through veins. Blood in arteries flows
from heart, and when reaches brain produces animal spirit. Only contact between the two
flows were through pores in heart. Lungs bring air to heart, and discharge smoky vapors
produced when air reached the invisible fire in the heart (like a hearth).
USE OF MEDICATION
- large scale use of medicines derived from plants. prepared
prescriptions.
- Use of medicine often went to extremes. Mixed and blended agents
whose properties classified according to 4 humors and 4 qualities (hot, cold, dry, moist).
i.e. a hot illness required a cold drug.
- Expanded number of ingredients to more than 70 in theriac, an
ancient medicine originally used for snake bites, but whose use was expanded for use
against all poisons, pestilence, black death. Called theriac (means wild beast) after
viper flesh was added to it . By middle ages, more than 100 ingredients. Productions took
months, and should be aged for years. Its use lasted until late 19th century. Popular
partly since it had opium in it.
CONCEPT OF BLOOD
- ingested food digested then transformed in form of "chyle"
to liver, where it is turned into blood
- in liver, bile charged with "natural spirit" necessary for
life function
- blood leaves liver by veins to carry nourishment to body
- some of blood move through arterial vein (our modern pulmonary
artery) to lung, where gives up impurities and waste to air
- some blood enter right ventricle of heart, which passed to left
ventricle of heart through small holes, where it mixes with air that comes form lungs by
venal vein (our modern pulmonary vein) and becomes charged with "animal spirit",
changing in color from dark red (which we know is deoxygenated blood) to bright red blood
(which is oxygenated blood).
- This new blood pass through arteries to body
- An additional system, arising in brain, is source of "animal
spirit" which is sent out through hollow nerves.
WHY DID HIS WORK LAST SO LONG?
- middle ages produced wish for authority and absolutes
- teleological reasoning was in accord with the teachings of the
Christian church.
- his encyclopedic writing were great resources for training and
information
- practitioners that followed him elevated his status
- remained unchallenged until Vesalius, 16th century, who shook
foundations of authority in Renaissance.
GRAPHIC:
PHYSIOLOGY OF GALEN
MEDICINE AND THE RISE OF CHRISTIANITY
- Early Christians thought disease associated with sin; response was to
suffer, and the only cure was through Grace.
- When 2nd "coming" didn't occur, became more concerned with
health and disease
- touching became important in healing, mimicking described healings of
Christ.
- As church became more structured, tried to extend Christ's healing
mission so develop facilities for care of diseased. Hospitals developed, unlike those used
on battlefields, since they cared for long-term support of diseased and poor.
MEDICINE AND THE DARK AGES
- After fall of Rome to Goths in 476, and decline of Roman culture and
influence, "ancient" knowledge in science and medicine knowledge decline in
importance with concomitant increase in power of Church. People took knowledge on
"faith" with less emphasis on learning and own observations. Increased interest
in magic and mysticism.
- Return to belief in supernatural cause of disease, with cure by
exorcism, healing plants, blood letting
- Many area monastic communities became place for treating illnesses. St.
Benedict encourage care of sick, but cure only through God. Effects continue for
500 years. He recommended study of Hippocrates and Galen and preserved medical literature,
but didn't use it. Techniques of surgery lost replace by cauterization. Pharmacology
abandoned and people used common herbalism, folk medicine.
MEDICINE AND THE RISE OF ISLAM (Muhammad 570-632)
First five centuries of Christian era, much of Greek scholarship
lost. However, in the 7th century, the new religion of Islam started to preserve remains
of ancient scholarship and bring it back into Europe. Arabs had great respect for
learning, and became the vessel for ancient Greek/Roman learning. They translated and
extended the ancient knowledge, especially in philosophy, math and science.
- after Muhammad, Islam swept in Mid East and into Africa, Spain, and
parts of France, with earliest center in Baghdad. Aristotle, Hippocrates, and Galen
translated into Arabic. In Egypt, hospital of Cairo probably most advance in the Christian
or Moslem world.
- Arabist (Arab, but also Persians, Jews, and Christians who lived or
moved to region) responsible for establishment of pharmacy and chemistry as science. New
drugs became part of pharmacy. New methods to extract drugs and preparing them developed -
distillation, crystallization, solutions, sublimation, reduction, calcination. Modern
chemical terms derived from Arabic include alkali, alcohol, as well as older terms of
elixir and syrup.
- strong emphasis on clinical instruction, and described many disease
much better than predecessors.
- development of efficient hospitals far exceeded anything before its
time.
- Disease thought to be punishment from Allah. Seek miracles but also
divine help through physician. Since there was afterlife, and a vital life spark remained
in dead body, forbade dissections. Relied on Galen for knowledge of anatomy.
- Diagnosis based on: behavior, excrement, swellings, kind and location
of pain, characteristics of urine. Properties of pulse noted. Astrology had role since
influence of stars part of science.
- Surgery held on low regard. Muslim lab forbade dissections.
Cauterization most common technique. Use sponge filled with narcotic-like substances for
anesthesia. Did do cataract surgeries.
- Wide use of drugs.
- Most widely influential Arab in medical field was Avicenna
, 980-1037

Equal to Galen in both Islam and Christian world. Boy prodigy.
Principle contribution as compiler and commentator. Wrote 100 books, most famous The
Canon, which was basis for much medicine, even until mid 17th centuries in Christian
Universities. Tried to reconcile Galen with Aristotle.
MEDICINE AND THE RISE OF UNIVERSITIES
Real medical education established in new universities such as
Salerno, Monpellier, and Paris:.
- Salerno (southern Italy): supposedly founded in 9th century by 4
physicians (a Greek, Latin, Hebrew, and Arab). Open to women as well, most famous
legendary Trotula, author of book on obstetrics. Still used Galen anatomy, and physicians
dissect animals, especially pigs. 1224, emperor stated that all candidates for licensure
must be examined by masters at Salerno after studying logic for 3 yr., medicine and
surgery for 5, and practicing under a master for 1.
- Surgery: revival of dissections of human cadavers to determine cause
of death and learn about nature of disease.
MEDICINE AND THE MIDDLE AGES
Western Europe emerged from dark ages about 1000 AD, but still
another 500 years before middle ages ended. French culture with combination of faith,
feudalism, and chivalry model for Europe. Each person had role/function. Mediterranean
culture in disarray.
- Role of Church - Aquinas: 1225-1274:
Such a supporter of Aristotelian thought that his influence lasted through middle ages.
Didn't believe Nature did anything unnecessarily; hence everything in Nature had simple
explanation. Thought change occurred when the essence of things become expressed (such as
an acorn becoming an Oak tree). No being can become something without the inherent
potential to do so. Hence all other changes come from God, the power source of all change.
Lead to his doctrine of effective causation, which was foundation of Scholasticism, where
supernatural had supremacy over Nature. Suppressed independent thought for the rest of the
middle ages. Even though Church controlled universities, monastic medicine decline, and
hospitals control shifted form the Church to cities.
Aquinas: – rediscovered Aristotle,
Christianized his philosophy – basis for later Middle Ages. Summa Theologica. Aim to
show harmony btw Aristotle and Christianity. Ethics 2 dimensions: natural and theological.
Natural ethics (as from Aristotle) – development of reason and practice in living
morally and leads to earthly happiness.. Theological ethics – achieve virtues of
faith, hope, and charity through grace which lead to eternal life. Believe in natural lab:
divine law as written in hearts of people. – which discovered by reason and
cultivated by conscience. Since allow to turn to secular knowledge without guilt, pave way
for more scientific view of world
Natural Law - dominant theory in Christian thought
- world is rational order with values and purposes built into its nature (from Greek
thought) - teleological. Aristotle 4 ?: What is it; What's it made of? How did it come to
exist: What is is for? (Plants for animals, etc - Nature make all ultimately for man. -
anthropocentric. Christians liked but add God - God made things for purpose, including
values
- (corollary) Laws of nature describe how things are and ought to be. Drought natural
evil; Some ways behaving natural (derived from nature), other unnatural and morally wrong;
supports beneficence but others more contentious as well - sex zb.
Moral judgments dictates from laws of reason which grasp since God, author, made us
rational. Religious believer no special access to moral truth. all must reason. Aquinas
most influential natural law theorist: moral life lived "according to reason"
(Summa Theologocia) Conscience as voice of God (but not apparent to disbeliever)
- Medicine: physicians available to upper classes, masses rely on folk
medicine with concoctions and magic. Establishment of guilds which people would join (such
as Artist and Physician guilds)
- Crusades: generated hospitals and orders for healing. the
"hospitalers" assisted victims of epidemics resulting in part from return of
Crusaders.
- Disease:
- leprosy : found in low levels before Crusades, but after return epidemic levels.
Disease include many skin diseases, of which some were contagious. Victims ostracized, had
to wear certain clothes, worship separately.
- Black Death (pneumonic and bubonic plaques) :
Known previously but reappear in 1347 as it moved west from India and SW Russia with
Crusaders. Most of them died on route home. When reached Italy, started an epidemic which
spread all the way to Greenland. Within 1 year, 1/4 of European population dead. Higher in
certain locales (Marseilles - 4/5). Start general dissolution of society and end of middle
ages, as well as growing nationalism, influx into cities. People lost faith in both
government and church, adopting new beliefs/trends to replace them. Astrology, black
magic, search for saint relics, search for magic holy waters common.
- Physicians and Treatment: major development in Middle Ages was the
regulation of physician training, ideas of contagion, and the care of very ill and
unwanted. Diet important, drugs heavily used, especially plants. Mysticism more prevalent,
even as science used more. Astrology and devil used to explain disease. Remedy often
exorcism. Combination of science and mysticism; Treat imbalance of humors by bloodletting.
Surgeons branch out into two groups, those with greater education and the barber, who
performed tooth extractions, minor operations, and setting bones.
MEDICINE AND THE RENAISSANCE
Many factors drove the changes that became the Renaissance: a desire
to return to cultural ideas of ancient Romans and Greeks, the invention of the printing
press which decrease the cost of books which also became available in the vernacular, the
development of trade resulting from the Crusades and the development of local industry,
leading to a money economy, and the discovery of sea routes to India and America. After
Turks capture Constantinople in 1453, a large influx of Greek scholars into northern Italy
occurred. The Universities there (Bologna, Padua, etc.) attracted scholars from all
Europe. The best publishing houses developed in Florence and Venice. Northern Italy was
also the western end of the Silk Road, from which goods were transported to and from
China. The arriving Greek scholars de-emphasized Aristotle, Galen, and Scholasticism,
while supporting Hippocrates and open observation. With the 17th century and the Age of
Scientific Revolutions, major intellectual shifts occurred. People shifted to asking how
things happen rather than why they occur? Speculation was replaced with experiment, and
interpretations became mechanical. Three forces had to be dealt with:
- Aristotle, where chance not a factor, but where purpose was not
obvious and could be determined
- Galen, where search for facts, clouded by preconceptions and
misconceptions, was important. He disrespected authority except Hippocrates and himself.
His desired to see for himself was not used by his followers who blindly followed him.
- Paracelus, who opposed
Galen and Avicenna, and emphasized observation and experience, but whose own system was
astrological. He did presume the body to be a chemical machine, and popularized use of
minerals, not just plants in treatment.
PARACELUS: 1493-1541
- opposed the teachings of Galen and burned copy of the standard text
of the day, Cannon of Medicine, by Avicenna.
- lectured in vernacular (German), rejected organized religion and
classical scholarship.
- opposed the leading medical theory: disease is result of imbalance of
four humors (body fluids) - blood, phlegm, choler or yellow bile, and melancholy or black
bile - in relation to composition of the person. Implies that as many different illnesses
as people, and that disease not caused by agent and don't have specific anatomical
effects.
- in contrast, said disease caused by outside agent and have an effect
on a specific anatomical site.
- Traditional medicine treat disease by by causing sweat, purging,
bloodletting, inducing vomiting; Parcacelus tried to find specific substances to cure
disease.
- He produced new substances to treat disease as an alchemist, not as a
modern chemist., since he believed in three principles (salt, sulfur, and mercury) which
possessed spiritual properties.
- his work helped to turn alchemy away from turning elements into gold
into finding "drugs" to treat disease
- opposed build up of medical system based on anatomy and physiology;
opposed most organized schools of thoughts. .
VESALIUS - 1514-1564
- father of modern anatomical science; inaugurate reform of anatomy and
teaching
- 1537 taught surgery and anatomy at U. Padua, where he did dissections
himself rather than have surgeons do it
- 1539, judges allowed the dissection of corpses of executed criminals,
allowing him and others to make great progress in anatomy
- 1543, wrote De Humani Corporis Fabrica (on the Construction [fabric]
of the Human Body), with great plate illustrations.
- said that the body must be studies directly, not just by reading
- from his dissections, showed that Galen's anatomy of humans was based
on studies of animals and not accurate for humans
- tried to convince doctors and anatomist of inadequacy and errors in
Galen's work
- didn't have the impact of others since he did not attack Galen or his
doctrines directly; criticized only Galen's writing on anatomy and not the Galenic system
of medicine.
- for example: didn't finds pores in the heart which allow blood to
ooze dropwise from the "arterial vein" (or modern pulmonary artery, which takes
deoxygenated blood from the heart to the lung) into the "venal artery"
(pulmonary vein, which carries oxygenated blood from the lung to the heart.). [Remember,
veins except the pulmonary vein, carry deoxygenated blood from the body back to the
heart.] These pores, part of Galen's heart physiology, do not exist.
HARVEY: 1578-1657
- revolutionary in that his studies that showed blood is pumped by the
heart in a single, closed mechanical system through veins and arteries, and not in
imagined paths;
- established new basis of human anatomy and physiology, replacing 15
centuries of Galenic medicine .
- teacher included anatomist Fabricius, who discovered valves in veins.
- major book, De Motu Cordis (1628) describes circulation of the
blood., one of the most important works of the 17th century, revolutionized biological and
medical thought in 3 ways:
- firm establishment of experiment and observation to advance biology and life sciences
- introduced quantitative reasoning to study life processes
- ideas of blood circulation
- recognized that vein valves discovered by Farbricius allow only one
way flow of blood through veins.
- valves suggests pump, so Harvey treated the heart as a pump.
- heart acts by contracting (when blood is expelled) and expanding.
(when blood is pulled into the heart)
- Harvey show: blood forced out of left ventricle into aorta, then into
arterial system, Blood return to heart though veins in right ventricle. Then moves to
right auricle and then through pulmonary artery to lungs. From there it flows back by
pulmonary vein into the left auricle , then the left ventricle. The cycle is then
repeated. Confirmed by experimentation, observation, dissection, correcting an error held
for 2000 years. Studies 80 different species.
- used mathematics to explain biology. Determined capacity of heart,
(volume pumped per minute) which when multiplied by the number of beats per minute gives
the total volume pumped by the heart in one minute. He fund that 164 lbs. of blood is
pumped from the heart into the arteries each hour, which is "more blood than the
ingested food can supply, or that all the veins together at any give time can
contain". Therefore blood flow had to be in a circuit.
- Harvey did not know about capillaries, which were discovered later by
Malpighi.
- revolutionized biology but not necessarily the practice of medicine.
DESCARTE - 1637
- believed true science would advance medicine and technical arts;
would be a utility to life and allow one to master nature.
- argued that greatest principle of common sense: "All things
which we cleanly and instinctively conceive are true".
- From I think, therefore I am (cogito ergo sum) (i.e. an intuition),
that a consciousness of his own thinking proved his existence, he argued for existence of
God.
- God made two classes of substances:
- thinking substances or minds and
- extended substances or body
- part of blood was a fluid called animal spirit, that came into
contact with thinking substances in brain, and flowed over channels of nerves to animate
muscles.
BASIS OF MODERN MEDICINE
Germ Theory of Disease - Infectious Disease
The Emergence of Modern
Medicine in America in the 19th Century:
Post-Genomic Medicine
Guides to Use of Medical
Literature
In analyzing the results of a published medical study, you must have
confidence that the results of the study are valid - i.e. that they conclusions follow
directly from the observations - and that you understand the results of a treatment.
Consider an article published in the Journal of the American Medical Society (1993)
270, pg 2598-2601 entitled, "How to Use an Article About Therapy and Prevention"
and "How to Use an Article About Harm", JAMA (1994) 271, pg 1615-1619.
Are the results of the study valid?:
Do the results represent an unbiased estimate of a treatment's positive
effects or of a potential toxic effect of exposure or have they been influenced in some
systematic fashion to lead to a false conclusion? Several factors must be
considered?
A. Where there clearly identified comparison groups (experimental
vs. control) that were similar with respect to important determinants of Outcomes other
than the one of interest - i.e. the dependent variable? Three kinds of trials
have been used in such assessments:
- Randomized Control Trials (RCT) - An experiment in
which patients are assigned in a random fashion to the experimental group (i.e.
given a treatment of unknown efficacy) or control groups (i.e. for a treatment study,
given a placebo or standard therapy). RCT are rarely used for harm studies.
The power of RCT stems from the fact that patients are randomly selected which raises the
probability that experimental and control groups are identical except for the experimental
treatment.
- Cohort Studies - Used when it is impossible or
unethical to perform a RCT. In this study the investigators identifies two groups of
people: one using a particular treatment or exposed to a potentially harmful agent
and another not using the treatment or not exposed. The participants are followed to
determine difference in effects of treatment or exposure to harm with time. An
example would be to select men (or women) exposed to toxins in a given industry and
assess the health status of their children. Alternatively, cohort studies can be
done when the outcome is infrequent. Since the people in the study are not randomly
picked (since they pick themselves or are picked by doctors), members of the exposed group
may be different in other characteristics from the control. Investigators must
show that the groups are similar (except with respect to the dependent variable) or use
statistics to adjust for any differences. (For example, a study for GI bleeding in
people who take non-steroidal anti-inflammatory drugs (NSAID) should be controlled for
age, since increased age as well as increased use of NSAID are associated with GI
bleeding.) Even then other confounding variables might be associated with the
effect, such as if illness for which people use NSAID might cause GI bleeding
- Case Control - Used if outcome very rare or takes
long to develop. Investigators select people with specific outcome and then
chooses controls without that outcome but as similar in other ways to experiment group
(ex: identical in age, sex, weight, etc). then ascertain past exposure to therapy or
toxins in both groups. (For example, show association between DES intake by pregnant
women and development of cancer in their daughters.
- Case Reports - Do not use control groups and are
merely descriptive.
B. Were the exposures to potentially toxic agent and the
outcomes (of RCT and cohort studies) measured the same in both experimental and
control groups? For example, in a case control study, when asked about prior
exposure to solvents, patients in the experimental group (with leukemia) might be more
likely to recall and report previous exposure because of greater motivation (recall
bias) or greater probing by an interviewer (interviewer bias).
Exposures to potentially toxic agents should also be for a similar length.
In a cohort study, a three-fold increase risk of cancer in radiation workers might be
explained by a physician who was aware of the risk doing a more thorough assessment of the
patient which might detect disease earlier or disease that would ordinarily go undetected
(surveillance bias).
C. Were the patients available for follow-up for a sufficiently
long enough period of time? If patients are not available for follow-up, the
validity of the study might be compromised since that that remain available might have
different prognosis than those who aren't available.
D. Were patients analyzed in the groups to which they were
randomized? If someone in a experimental group taking a drug become
noncompliant and don't take the medication, they should not be excluded from the analyses.
Often these patients fared worse. Excluding them (while leaving similar
patients in the control group) leaves behind those who might have gotten better, and
removes the unbiased nature of the RCT.
E. Were patients, clinicians,
and study personnel "blind" to treatment? If a patient knows they are
on a new therapy, they will have an opinion about its efficacy, as will the practitioner
and others involved in the study. The opinions can systematically distort aspects of
the treatment, reporting of outcomes, and introduce bias of the medical personal into the
assessment of the treatment efficacy. Best way to avoid is performing a double-blind
study (for drug studies, using a placebo for the control group and target drug for
the experimental group).
BIBLIOGRAPHY
The information for the time line and descriptions
come directly and often verbatim from a variety of sources, including:
Butterfield, H. The Origins of Modern Science.
Free Press, 1965
Lyons, A. and Petrucelli, R. Medicine: An
Illustrated History. Abradale Press, 1987.




