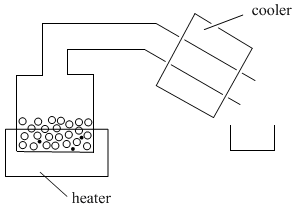
PM3.3. Distillation
Distillation is a method of purifying organic compounds. It takes advantage of the fact that two different compounds probably have two different boiling points. Suppose two different liquids are present in a homogeneous mixture (they are completely miscible, or they mix completely together, like water and alcohol). If they have two different boiling points, one of the compounds will evaporate before the other one does. The more volatile compound (the one that evaporates easily) will leave the less volatile compound behind.
Distillation is probably a familiar word. Distilleries are factories that produce alcoholic beverages, such as whiskey, from the fermentation of grains, such as corn or rye. At the heart of the distillery is a large distillation apparatus. This "still' is used to purify the alcohol/water mixture by evaporation, leaving behind most of the other components of the grain. That process is slightly more complicated than the one described here, but it is similar.
In distillation, a mixture is put into a container and the container is placed on a heat source. A tube leads away from the container. Sometimes this tube is cooled by cold, running water or ice; sometimes it is just left to cool in the air. There is also a second container to collect the liquid that distills.

As the container or "pot" is heated, the volatile liquid begins to evaporate. Molecules enter the gas phase and begin to float through the tube leading away from the pot. In the lab, the tube above the pot is called the "still head".
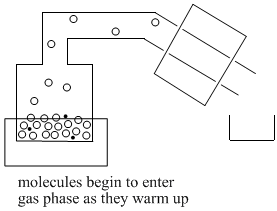
Once molecules get past the still head, they reach the "condensor". Often the condensor is cooled by cold water that flows along outside of it. When molecules reach the cold condensor, they give up some heat energy and condense back into the liquid phase.
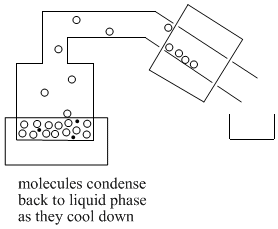
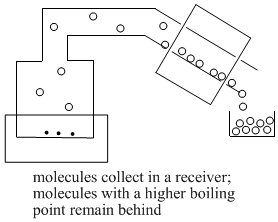
As enough molecules condense into the liquid phase, they begin to form drops of liquid. This liquid collects in a "receiver flask".
Problem PM3.1.
Decide whether the following mixtures are good candidates for distillation.
a) You have a sample of pentane (mp: -129°C; bp: 36°C) contaminated with 1-octanol (mp: -16°C; bp: 195°C).
b) You have a sample of 1-hexanol (mp: -50°C; bp: 145°C) contaminated with 2-hexanol (mp: -16°C; bp: 140°C).
c) You have a sample of benzene (mp: 5°C; bp: 80°C) contaminated with benzoic acid (mp: 122°C; bp: 250°C).
d) You have a sample of 2-butanone (mp: -86°C; bp: 80°C) contaminated with toluene (mp: -95°C; bp: 111°C).
e) You have a sample of naphthalene (mp: 78°C; bp: 218°C) contaminated with 2-naphthol (mp: 120°C; bp: 285°C).
f) You have a sample of 1-hexanol (mp: -50°C; bp: 145°C) contaminated with 1-hexene (mp: -140°C; bp: 63°C).
This site is written and maintained by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's University (with contributions from other authors as noted). It is freely available for educational use.

Structure & Reactivity in Organic, Biological and Inorganic Chemistry by Chris Schaller is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported License.
Send corrections to cschaller@csbsju.edu
This material is based upon work supported by the National Science Foundation under Grant No. 1043566.
Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Back to Structure & Reactivity