Reactivity in Chemistry
Aliphatic Nucleophilic Substitution
NS5. Regiochemistry
Regiochemistry is the term for where changes take place in a reaction. It can be another indication of how the reaction occurred.
In aliphatic nucleophilic substitution, the answer seems pretty obvious: the reaction takes place at the electrophilic carbon, the one attached to the electronegative halogen. That's where the leaving group is. When the leaving group is replaced, that's where the nucleophile will be. That's always true in an SN2 reaction, but it isn't always true in an SN1 reaction.
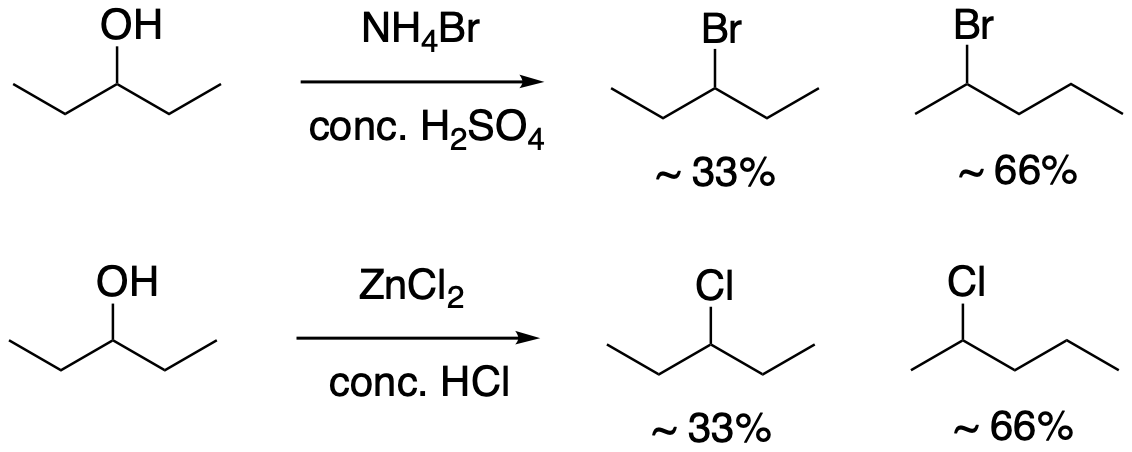
Under some circumstances, unexpected changes occur. The following two reactions are examples of such surprises. Notice the leaving group appears to be an OH group this time. A C-O bond is polar, so the attached carbon is potentially electrophilic. However, a hydroxide ion isn't as stable as a chloride, bromide, or iodide ion, so it isn't such a good leaving group. That's why these reactions are in acidic media. The strong acid protonates the OH group to make water, which is a better leaving group that hydroxide would be. These reactions happen to take place via an SN1 mechanism.

Figure NS5.1. Rearrangement of the electrophile structure in an SN1 reaction.
Notice there are two different products in each reaction. So, the regiochemistry of this reaction may be more complicated than we thought.
What is happening in these two reactions? In one of them, a bromine from the ammonium bromide, NH4Br, is replacing the OH group. In the other reaction, a similar thing is happening with a chlorine from the hydrochloric acid. There is also a second product in which the bromine or chlorine is in the wrong place. If you look carefully, it seems as if the halogen has switched places with a hydrogen atom next to the electrophilic position. It doesn't seem like that hydrogen atom could come off very easily.
This sort of behaviour is characteristic of carbocations. It is called a rearrangement, in which part of the molecule unexpectedly switches places.
Again, one of the products forms in a simple enough way.
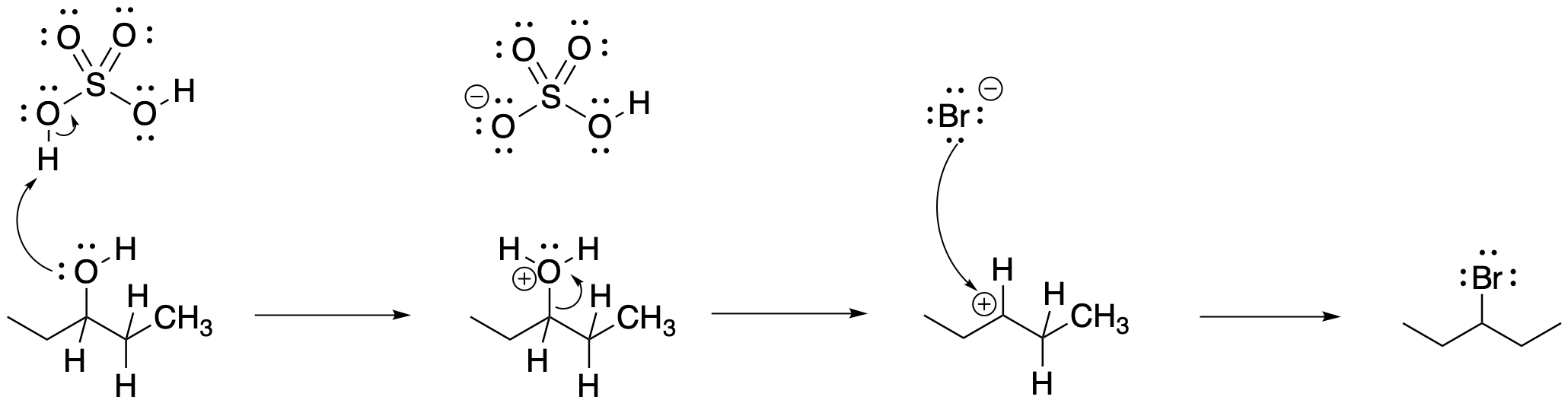
Figure NS5.2. An SN1 reaction of an alcohol, assisted by strong acid.
The formation of the other product involves a "1,2-hydride shift". In this event, a hydrogen anion hops from one carbon to the next, leaving a cation where it used to be.
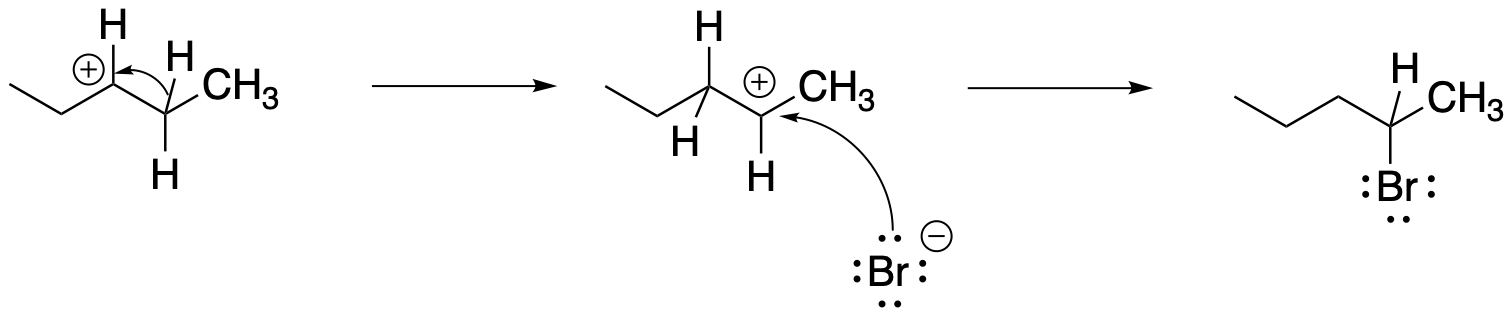
Figure NS5.3. Rearrangement via a 1,2-hydride shift during an SN1 reaction.
Rearrangements like this are very common in substitution reactions of alcohols, as long as the mechanism is SN1. They can also happen when alkyl halides are subjected to substitution reactions. All that is required is for the reaction to pass through a cationic intermediates. No matter the starting material or leaving group, rearrangements can occur during SN1 reactions.
Problem NS5.1.
Draw the reaction above with all the hydrogens drawn in the structures, to confirm the formal charges and the positions of the hydrogens.
Problem NS5.2.
Explain the observed product ratios in the above reaction of 3-pentanol with ZnCl2 and HCl.
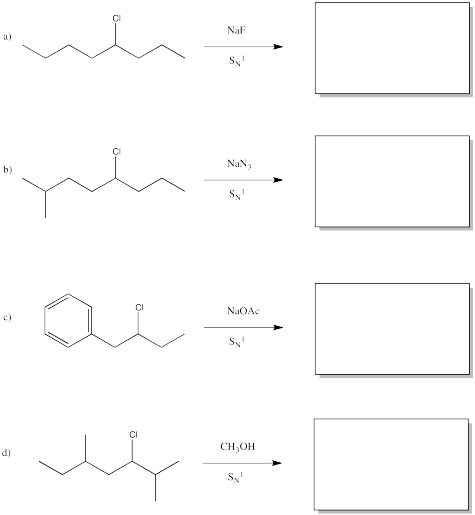
Problem NS5.3.
Predict the products of the following reactions.
The hydride can hop one carbon away because of the proximity to the empty p orbital with which it can overlap and form a new bond.
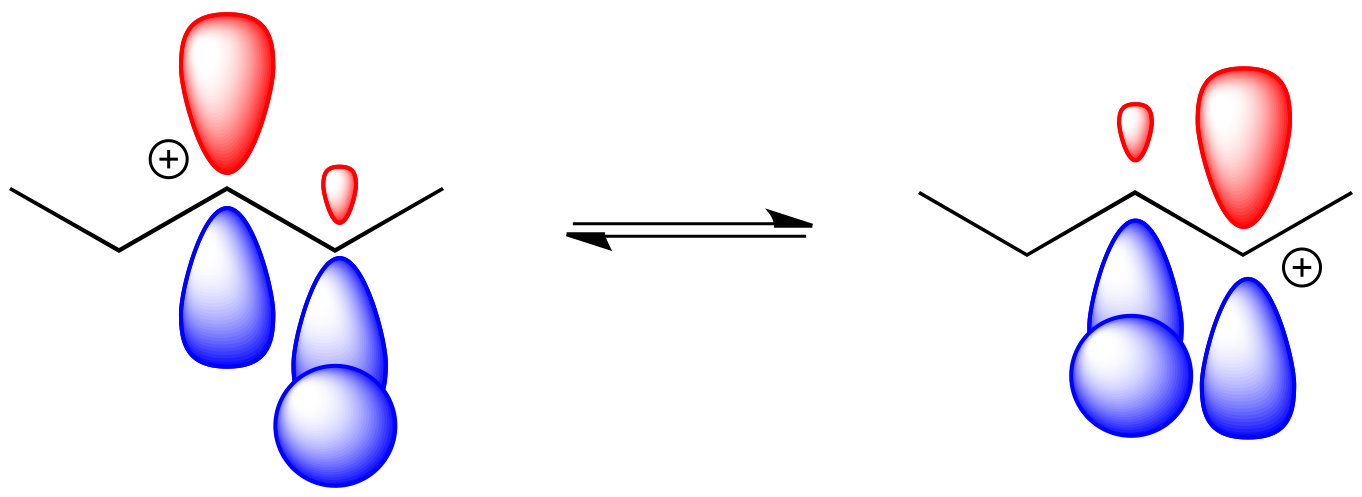
Figure NS5.4. Orbital overlap during a 1,2-hydride shift.
The barrier for a hydride shift is not very high, provided a carbocation is available on the very next carbon. As a result, an equilibrium between cations is established pretty quickly. Below, there is an equilibrium between two secondary cations on the 2-methylpentyl skeleton.
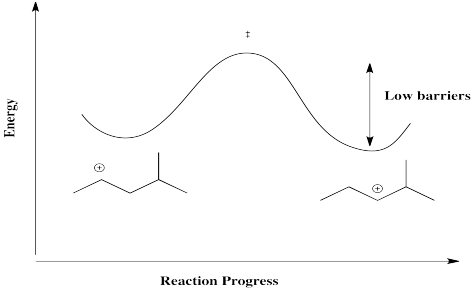
Figure NS5.5. Reaction progress diagram for a 1,2-hydride shift.
However, that particular structure has another possible cation that is more stable. Once a tertiary cation forms, the hydride isn't likely to hop back.
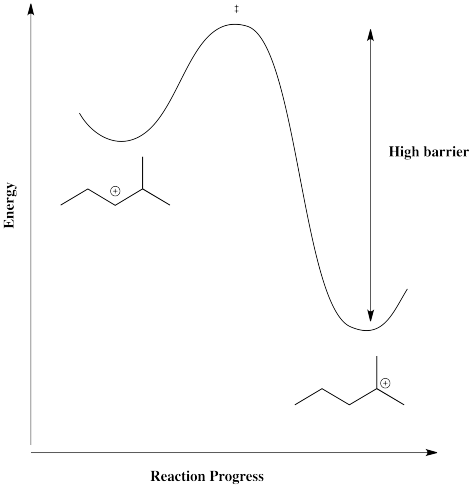
Figure NS5.6. A high barrier to return to a secondary cation from a tertiary one via a 1,2-hydride shift.
On the other hand, there is also a primary position. A hydride shift could give a primary cation, but that isn't likely to happen, because it would be too far uphill.
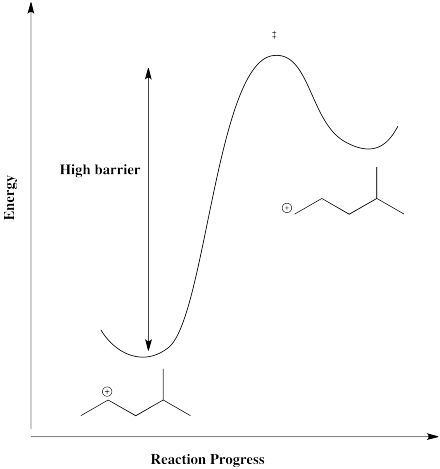
Figure NS5.7. A high barrier to form a primary cation from a secondary one via a 1,2-hydride shift.
Altogether, there is an energy surface linking several different possible cations. In this case, however, one tertiary cation would quickly dominate.
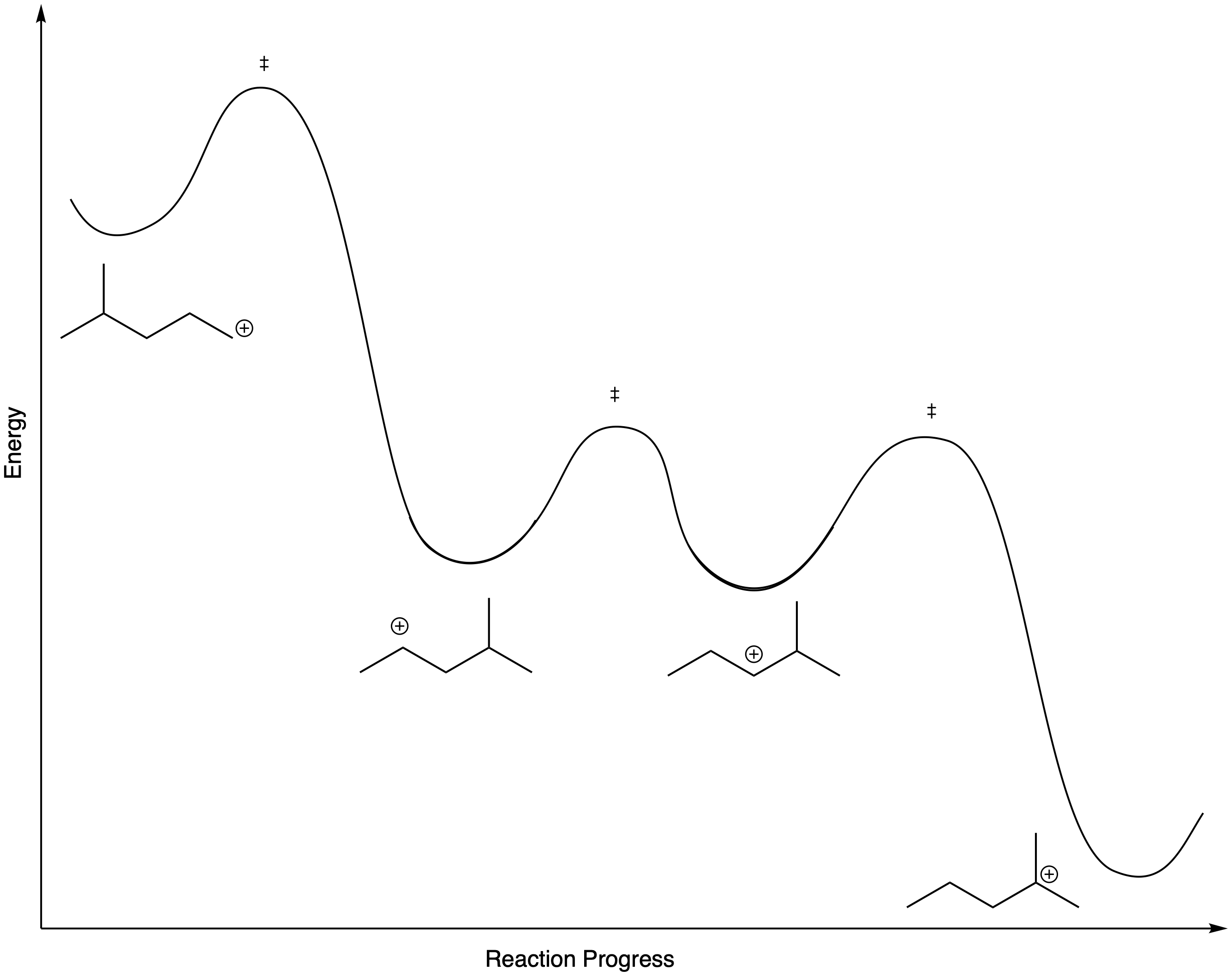
Figure NS5.8. The energy surface linking different 4-methylpentyl cations.
It isn't just hydride ions that are able to undergo 1,2-migrations. Alkyl anions (such as methyl, CH3-) and aryl anions (such as phenyl, C6H5-) can also undergo 1,2-shifts, rearranging to give stable cations.
For example, the following reaction is apparently just a substitution of a bromo group for a hydroxy group. The regiochemistry indicates a cation was formed, however, because the new group is found at the site of the most stable carbocation.
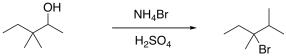
Figure NS5.9. Rearrangement of a carbon skeleton during an SN1 reaction.
The mechanism here involves protonation of the hydroxy group; the reaction takes place in strong, concentrated acid. The resulting cation is able to undergo a 1,2-methyl shift leading to a new carbocation. The bromide ion connects at that new position.
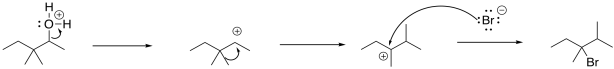
Figure NS5.10. A 1,2-methyl shift during an SN1 reaction.
This site was written by Chris P. Schaller, Ph.D., College of Saint Benedict / Saint John's University (retired) with other authors as noted). It is freely available for educational use.

Structure & Reactivity in Organic, Biological and Inorganic Chemistry by Chris Schaller is licensed under a Creative Commons Attribution-NonCommercial 3.0 Unported License.
Send corrections to cschaller@csbsju.edu
This material is based upon work supported by the National Science Foundation under Grant No. 1043566.
Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.
Navigation: