Biochemistry Online: An Approach Based on Chemical Logic

CHAPTER 9 - SIGNAL TRANSDUCTION
C: SIGNALING PROTEINS
BIOCHEMISTRY - DR. JAKUBOWSKI
04/16/16
|
Learning Goals/Objectives for Chapter 9C:
|
Estonian Translation √ by Anna Galovich
C13. Metabotropic Neural Receptors
You may have noticed above that some signaling molecules, whose effects are regulated by kinases (β-adrenergic and some olfactory signals by PKA and acetylcholine by PKC for example), are neurotransmitters. In the previous chapter section, we discussed how neurotransmitters can act as signals to open ion-specific, ligand-gated membrane channels, which change the transmembrane potential. In other words, the neurotransmitters gate the channels directly. Typical examples of channels directly gated by neurotransmitters are the acetylcholine receptor in neuromuscular junctions and the Glu, Gly, and GABA receptors in the central nervous system. These receptors are multimeric proteins. Receptors with direct gating of ion flow are fast, with activities that last milliseconds, and are used in eliciting behavioral responses.
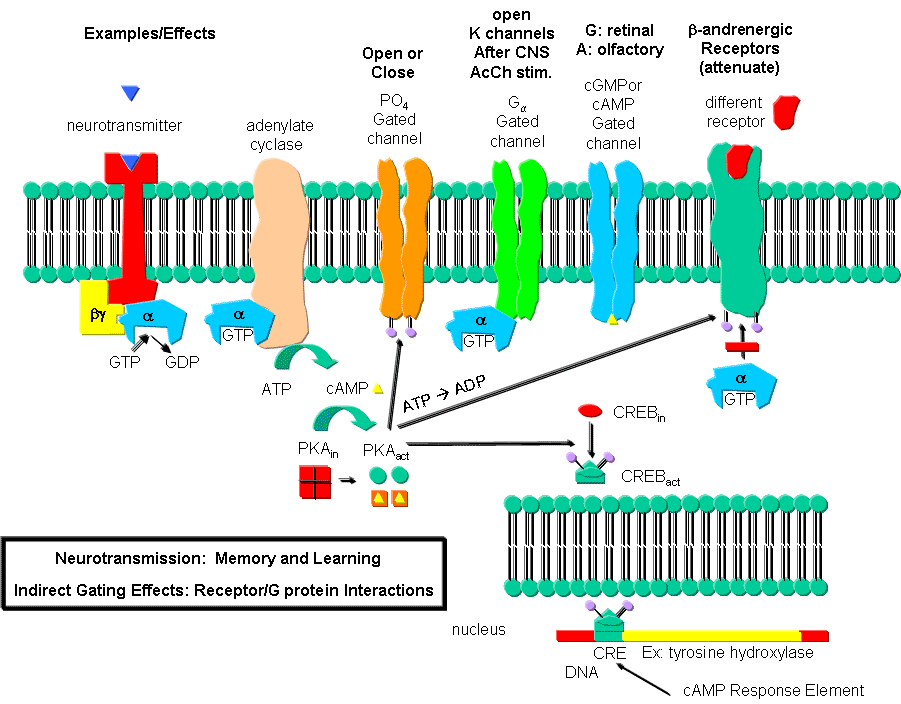
However, ion channels can also be gated indirectly when the neurotransmitter binds to its receptor and leads to events which open an ion channel that is distinct from the receptor. In this case, the occupied receptor communicates to an ion channel indirectly through a G protein. Example of this indirect gating of ion channels include the serotonin, adrenergic, and dopamine receptors in the brain. These receptors are classic single protein serpentine receptors with 7 transmembrane helices, and intracellular domains that can interact with G proteins as described above to increase second messenger levels (cAMP, DAG) in the neuron. These can either activate kinases in the cell, which phosphorylate ion channels to either open or close them, or can bind directly to the channel and modulate its activity through an allosteric conformational change. In some cases the G protein directly acts on the ion channel. These different ways are described below. In contrast to direct gating, receptors that indirectly gate ion channels have activities that are slow and last seconds to minutes. These receptors are usually involved in modulating behavior by changing the excitability of neurons and the strength of neural connections, hence modulating learning and memory. These changes can occur in many ways, summarized below and in the following link:
Figure: Neurotransmission: Gating through G Linked Receptors
![]() Animations:
Direct and Indirect Neurotransmitter Action
Animations:
Direct and Indirect Neurotransmitter Action
Phosphorylating ion channels: Receptors that act through a second messenger system can change ion channel activity by activating kinases which phosphorylate the channels. This may:
-
open the channel normally closed at the resting potential and produce an effect like gating.
-
close a channel usually open at the resting potential (such as nongated K channels which when closed would depolarize the cell and make it more excitable).
Gα interaction with ion channels:
-
the Ga subunit of the G protein interacts with K channels after stimulation of the CNS Acetylcholine receptor, opening the channel and hyperpolarizing the cell
Second messenger interaction with ion channels:
-
cGMP opens cation channels in retinal cells after activation of the photoreceptor by photons
-
cAMP opens cation channels in olfactory cells after activation of the olfactory receptor by odorants.
Second messenger effects on proteins other than ion channels (usually different receptors):
-
the β-adrenergic receptors are phosphorylated by PKA and PKC (activated by stimulation of a different neurotransmitter receptor linked through a G protein to produce increased levels of second messengers cAMP and diacylglycerol). When phosphorylated, the β-adrenergic receptor, itself activated through G protein) can't bind Gs. This in affect attenuates the response of the β-adrenergic receptor to its own neurotransmitter which leads to desensitization to that signal.
Second messenger regulates gene expression:
-
cAMP activated PKA can phosphorylate an inactive transcription factor in the cell, which then can bind to a section of DNA called the cAMP Response Element (CRE), which is upstream of certain genes, leading to the transcription of the genes. The transcription factor is called CREB for cAMP Response Element Binding protein. Example: tyrosine hydroxylase (a monooxygenase) is involved in the synthesis of epinephrine and norepinephrine. The activity of this protein is increased when it is phosphorylated by PKA. Hence its activity can be increased quickly by this modification of the already present protein. If an animal is subjected to severe or long term stress (cold or immobilization), presynaptic cells with norepinephrine will be stimlulated to release the neurotransmitter. This requires continual synthesis of the neurotransmitter by the presynaptic cell. The increase in synthesis of this neurotransmitter is caused by the presynaptic cell being stimulated by another neuron, whch leads to increased levels of cAMP, and ultimately activation of CREB which increases transcription of the hydroxylase gene.
Caffeine
Caffeine clearly produces a state of arousal in the central nervous system. High levels appear to block the binding of an inhibitory neurotransmitter, adenosine, to the A2A adenosine receptor. In the absence of caffeine, adenosine levels rise during the day, which promotes interaction with its receptor, leading to increasing sleepiness and lack of concentration. When adenosine binds normally to its receptors, it activates the adenylate cyclase cascade, which activates PKA, leading to changes in phosphorylation state of many proteins inside the cell, including protein phosphatase (2A). These changes inhibit neural firing. Caffeine blocks these changes.
Navigation
Return to Chapter 9C. Signaling Proteins Sections
Return to Biochemistry Online Table of Contents
Archived version of full Chapter 9C: Signaling Proteins

Biochemistry Online by Henry Jakubowski is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.