Biochemistry Online: An Approach Based on Chemical Logic

CHAPTER 5 - BINDING
D: BINDING AND THE
CONTROL OF GENE
TRANSCRIPTION
BIOCHEMISTRY - DR. JAKUBOWSKI
Last Updated: 03/30/16
|
Learning Goals/Objectives for Chapter 5D: After class and this reading, students will be able to
|
D4. DNA Binding Sites
Since RNA polymerase must interact at the promoter site of all genes, you might expect that all genes would have a similar nucleotide sequence in the promoter region. This is found to be true for both prokaryotic and eukaryotic genes. You would expect, however, that all transcription factors would not have identical DNA binding sequences. The sequences of DNA just upstream of the start site of the gene that binds protein (RNA polymerase, transcription factors, etc) are called promoters. The table below shows the common DNA sequence motif called the Pribnow or TATA box found at around -10 base pairs upstream from the start site, and another at -35. Proteins bind to these sites and facilitate binding of RNA polymerase, leading to gene transcription.
Prokaryotic Promoter Sequences
|
Promoter |
-35 Region |
Spacer | -10 Region | Spacer | RNA start |
| trp operon | TTGACA | N17 | TTAACT | N7 | A |
| tRNAtyr | TTACA | N16 | TATGAT | N7 | A |
| λP2 | TTGACA | N17 | GATACT | N6 | G |
| lac operon | TTTACA | N17 | TATGTT | N6 | A |
| rec A | TTGATA | N16 | TATAAT | N7 | A |
| lex A | TTCCAA | N17 | TATACT | N6 | A |
| T7A3 | TTGACA | N17 | TACGAT | N7 | A |
| consensus | TTGACA | TATAAT |
A similar sequence is found in eukaryotes (consensus TATAAA) located about 25 nucleotides upstream from the transcription start site. It is called the Goldstein-Hogness box.
![]() Jmol:
Updated TATA Box Binding Protein
Jmol14 (Java) |
JSMol (HTML5)
Jmol:
Updated TATA Box Binding Protein
Jmol14 (Java) |
JSMol (HTML5)
In addition, in eukaryotes, sequences further upstream called response elements bind specific proteins (such as CREB or cyclic AMP response element binding protein) to further control gene transcription.
Eukaryotic Response Elements (RE)s
|
Regulatory agent |
Module |
Consensus | DNA bound | Factor | Size (daltons) |
| Heat Shock | HSE | CNNGAANNTCCNNG | 27 bp | HSTF | 93,000 |
| Glucocorticoid | GRE | TGGTACAAATGTTCT | 20 bp | Receptor | 94,000 |
| Cadmium | MRE | CGNCCCGGNCNC | . | ? | . |
| Phorbol Ester | TRE | TGACTCA | 22 bp | AP1 | 39,000 |
| Serum | SRE | CCATATTAGG | 20 bp | SRF | 52,000 |
| Antioxidant | ARE | GTGACTCAGC | |||
| Pheromone (fungus) | ACAAAGGGA | ||||
| Hypoxia | HRE |
CCACAGTGCATACGT GGGCTCCAACAGGTC CTCTCCCTCCCATGCA |
Hypoxia Inducible Factor | 826 aa | |
| Peroxisome Proliferator Activated Receptor (PPAR) | PPRE | aGG_CAAAGGT(CG)A | PPAR | 59,000 | |
| Steroid (general) (progesterone, androgen, mineralcorticoids, glucocorticoids | AGAACAxxxACAAGA (inverted repeat) |
![]() Eukaryotic
Promoter Database (EPD) - annotated non-redundant collection of
eukaryotic POL II promoters, for which the transcription start site has been
determined experimentally. Link to
article
describing EPD and its use.
Eukaryotic
Promoter Database (EPD) - annotated non-redundant collection of
eukaryotic POL II promoters, for which the transcription start site has been
determined experimentally. Link to
article
describing EPD and its use.
In addition to promoters, there are other DNA sequences that are more distal to the transcription start site than the proximal promoter that affect gene transcription. These include enhancers, silencers and insulators. The figure below shows how an enhancer might affect gene transcription by bringing distal DNA (perhaps thousands of base pairs away from the start site of transcription) close to the promoter for a gene. One could easily imagine how a silencer would work in an analogous fashion.
Figure: Enhancers of Transcription
Insulators are needed to prevent an enhancer (or silencer) from activating (or inhibiting) another nearby gene that should be affected. They are sequences between the enhancer (or silencer) and the promoter or a gene or a cluster of genes. Eukaryotic insulators have a CCCTC nucleotide site which bind proteins called CCCTC binding factors (CTCF).
The following figure and captions, reprinted from Nature with permission (as shown in the figure) shows a more detailed description of a simple eukaryotic transcriptional unit that controls gene transcription.
Figure: Eukaryotic promoters and regulatory regions
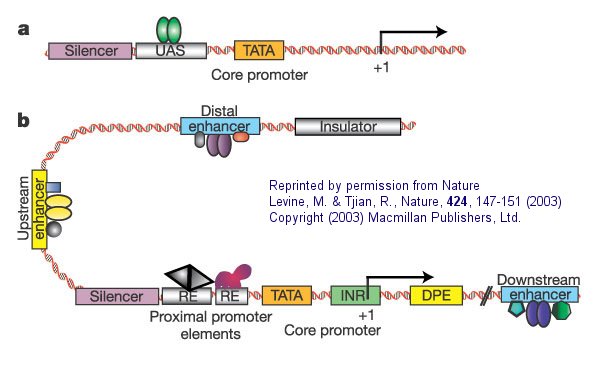
a. "Simple eukaryotic transcriptional unit. A simple core promoter (TATA), upstream activator sequence (UAS) and silencer element spaced within 100�200 bp of the TATA box that is typically found in unicellular eukaryotes".
b. "Complex metazoan (belonging to the kingdom Animalia) transcriptional control modules. A complex arrangement of multiple clustered enhancer modules interspersed with silencer and insulator elements which can be located 10�50 kb either upstream or downstream of a composite core promoter containing TATA box (TATA), Initiator sequences (INR), and downstream promoter elements (DPE)".
The following figure and caption, again taken with permission from Nature, shows a more detailed view of a multi-subunit general transcription apparatus, including tissue-specific and gene-selective subunits and transcription initiation complexes.
Figure: Eukaryotic multisubunit general transcription apparatus
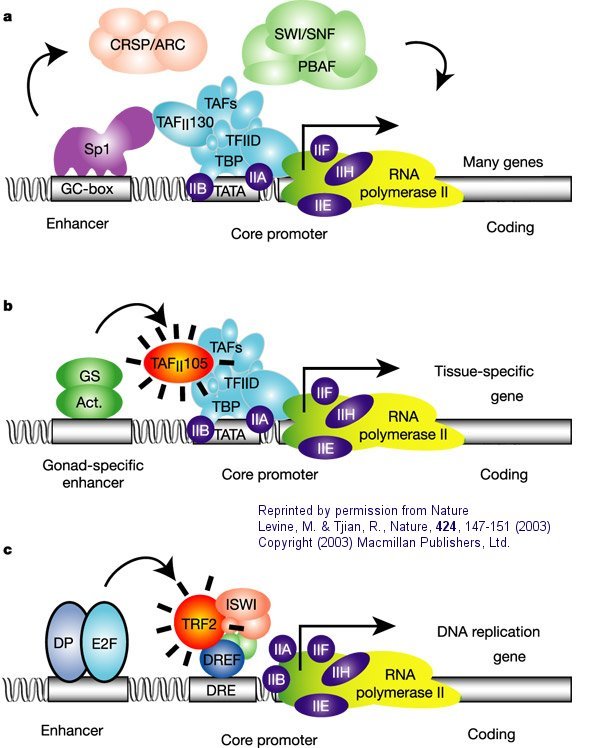
a. "The eukaryotic transcriptional apparatus can be subdivided into three broad classes of multi-subunit ensembles that include the RNA polymerase II core complex and associated general transcription factors (TFIIA, -B,-D,-E,-F and -H), multi-subunit cofactors (mediator, CRSP, TRAP, ARC/DRIP, and so on) and various chromatin modifying or remodeling complexes (SWI/SNF, PBAF, ACF, NURF and RSF)."
b, c. "Metazoan organisms have evolved multiple gene-selective and tissue-specific TFIID-like assemblies by using alternative TAFs (TBP-[TATA Binding Protein associated factors] such as the ovarian-specific TAF105) as well as TRFs (TBP-[TATA Binding Protein associated factors] related factors such as TRF2 in Drosophila and mice) to mediate the formation of specialized RNA polymerase initiation complexes that direct the transcription of tissue-specific and gene-selective programmes of expression." (Nature reference in figure above.)"
Proteins can interact specifically with DNA through electrostatic, H-bond, and hydrophobic interactions. AT and GC base pairs have available H bond donors and acceptors which are exposed in the major and minor grove of the ds DNA helix, allowing specific protein-DNA interactions.
Figure: AT and GC base pairs have available H bond donors and acceptors

![]() Jmol:
Updated Simple DNA Tutorial
Jmol14 (Java) |
JSMol (HTML5) (see last
selection buttons to see H bond donors and acceptors in the major grove.
Jmol:
Updated Simple DNA Tutorial
Jmol14 (Java) |
JSMol (HTML5) (see last
selection buttons to see H bond donors and acceptors in the major grove.
![]() Jmol:
Updated RNA Polymerase II/DNA/RNA
complex
Jmol14 (Java) |
JSMol (HTML5)
Jmol:
Updated RNA Polymerase II/DNA/RNA
complex
Jmol14 (Java) |
JSMol (HTML5)
Gene Transcription, Proteolysis and Membrane Lipids
An interesting example illustrates how transcriptional control maintains the balance of lipids in biological membranes. The phospholipids and sphingolipids in membranes are extremely heterogeneous, owing to the diversity of head groups and acyl chain composition. Given this great diversity, it is remarkable the different cells are able to maintain the specificity of lipid types in different cells, in different membranes within cells, and within a given leaflet of a membrane (remember our discussion of lipid rafts). How can the cell regulate the type of lipids that it synthesizes? What controls the transcription of genes for lipid synthesis?
Regulation of transcription of these genes appears to be controlled by multidomain proteins that bind to sterol response elements in the DNA. The proteins, called Sterol Response Element Binding Proteins (SREBPs) are activated by proteolysis to release a transcription factor domain which migrates to the nucleus. Proteolysis of SREBP occurs in the Golgi by resident proteases. The SREBP in the Golgi is in complex with another protein, SREP cleavage-activating protein (SCAP), which facilitates movement of the SREBP to the Golgi from its site of synthesis in the endoplasmic reticulum. Lipid regulation occurs when fatty acids, cholesterol, or PL derivatives like phosphoethanolamine (from ceramide) inhibits proteolytic activation of the SREBP. Regulation depends on whether or not SCAP "ferries" SREBP to the Golgi. SCAP binds to SREP and transfers it to the Golgi membrane, but only when sterol levels are low. When cholesterol is high, it binds to the transmembrane domain of SCAP and prevents SCAP from interacting with SREP and transferring it to the Golgi.
Auxin, a major plant hormone that induces gene expression, also seems to activate transcription through proteolysis. When bound to its soluble cytoplasmic receptors, ubiquitin protein ligase SCFTIR1, it activate proteolysis of protein that inhibit transcription.
![]() Jmol: Updated
Auxin Receptor
Jmol14 (Java) |
JSMol (HTML5) Auxin Receptor
Jmol: Updated
Auxin Receptor
Jmol14 (Java) |
JSMol (HTML5) Auxin Receptor
Navigation
Return to Chapter 5D: Binding and the Control of Gene Transcription
Return to Biochemistry Online Table of Contents
Archived version of full Chapter 5D: Binding and the Control of Gene Transcription

Biochemistry Online by Henry Jakubowski is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.