Biochemistry Online: An Approach Based on Chemical Logic

CHAPTER 5 - BINDING
F: IMMUNE SYSTEM RECOGNITION
BIOCHEMISTRY - DR. JAKUBOWSKI
Last Updated: 03/14/13
|
Learning Goals/Objectives for Chapter 5E: After class and this reading, students will be able to
|
03/31/16
F1. Introduction to the Immune System
This chapter is under development. Some of it is collected from other chapters in Biochemistry Online. In this last chapter on binding, we will consider the daunting task faced by the immune system - to recognize all possible "foreign" molecules and react to them, either by targeting them for elimination, or, paradoxically, to recognize them but not react to them (a process called tolerance). The same can be said of "self-molecules". The immune system must recognize them but not respond to them, otherwise autoimmune disease might arise in which the body's powerful immune system targets self.
It is virtually impossible to give an in-depth description of the immune system in a short chapter. My goal is simply to illustrate how the immune system recognizes such a myriad of molecules and how, through signal transduction processes, it responds by becoming tolerant of the target, or promoting an immune response. To accomplish this impossible task, I will brief cover the innate and adaptive immune system and their differences, and how some cells (macrophages in particular) in the innate immune system and cells (B and T cells) in the adaptive immune response recognize and response to target molecules and cells. Emphasis will be given to recognition and sample signal transduction responses. Much of the info presented here comes from ideas presented in a fantastic book written by Lauren Sompayrac, How the Immune System Works. (2003, Blackwell Publishing. ISBN: 0-632-04702-X)
Three lines of defense protect us from the "enemies", foreign substances (bacteria, viruses and their associated proteins, carbohydrates, and lipids) collectively called antigens.
- physical barriers of cells that line our outside surface and our respiratory, GI tract, and reproductive system
- the innate immune system (IS) that all animals have. Composed of scavenger cells like macrophages (MΦ), neutrophils, dendritic cells, and natural killer cells (NK) that can move around the body through the blood and lymph systems and burrow into tissue to meet the enemy where they can engulf and destroy bacteria and "cellular debris". Macrophages start off as immature circulating monocytes which enter tissues by slipping through blood vessel walls, and in the process they differentiate into macrophages. There they lie in wait ready for the enemy.
- the adaptive immune system, which, as its name implies, can change and adapt to new molecular threats. This branch is better at dealing with viruses which do their damage inside of host cells.. The adaptive IS is comprised of B cells that make and secret protein antibodies that recognize specific foreign molecules, and T cells.
F2. B and T Cells
What do B cells and antibodies do? In fact, nothing too exotic. They can't kill anything. B cells make antibodies. Antibodies just bind to foreign molecules (proteins, carbohydrates, lipids, etc), which might neutralize their effects, such as binding to the hemagglutinin molecule of the influenza virus and preventing its entry into the cell. They also bind to foreign cells like bacteria which signals other host immune proteins and cells to come in for the kill. Antibodies are secreted by B cells, which also have a membrane bound form of the antibody on their surface. This antibody acts as a receptor which recognizes antigen and through a signal transduction process, helps to activate the B cell. Mature B cells (those that have seen the antigen before) can churn out lots of antibodies quickly. Different surface antibodies, as well as secreted antibodies, can recognize and bind to almost any kind of molecules. The first antibodies produced by the immune system are often of low affinity. Over time, high affinity (low Kd) antibodies are produced.
![]()
![]() Jmol:
Antibody
Recognition of Antigens
|
Immunoglobulin Structure
Jmol:
Antibody
Recognition of Antigens
|
Immunoglobulin Structure
What differentiates high and low affinity binding at the molecular level? Do high affinity interactions have lots of intramolecular H-bonds, salt bridges, van der Waals interactions, or are hydrophobic interactions most important? Recently, the crystal structures of a variety of antibody-protein complexes were determined in order to study the basis of affinity maturation of antibody molecules. It is well know that antibodies elicited on exposure to a foreign molecule (antigen) are initially of lower affinity than antibodies released later in the immune response. An incredible number of different antibodies can be made by antibody-producing B cells due to genetic mechanisms (combining different variable regions of antibody genes through splicing, imprecise splicing, and hypermutation of critical nucleotides in the genes of antigen binding regions of antibodies). Clones of antibody-producing cells with higher affinity are selected through binding and clonal expansion of these cells. Investigators studied the crystal structure of 4 different antibodies which bound to the same site (epitope) on the protein antigen lysozyme. Increased affinity was correlated with increased buried apolar surface area and not with increased numbers of H bonds or salt bridges.
Table: Characteristics of Antibody:Hen Egg Lysozyme Complexes(HEL)
| Antibody | H26-HEL | H63-HEL | H10-HEL | H8-HEL |
| Kd (nM) | 7.14 | 3.60 | 0.313 | 0.200 |
| Intermolecular Interactions | ||||
| H bonds | 24 | 25 | 20 | 23 |
| VDW contacts | 159 | 144 | 134 | 153 |
| salt bridges | 1 | 1 | 1 | 1 |
|
Buried Surface Area |
||||
| ΔASURF (A2) | 1,812 | 1,825 | 1,824 | 1,872 |
| ΔASURF-polar (A2) | 1,149 | 1,101 | 1,075 | 1,052 |
| ΔASURF-apolar (A2) | 663 | 724 | 749 | 820 |
Li,Y. et al. Nature: Structural Biology. 6, pg 484 (2003)
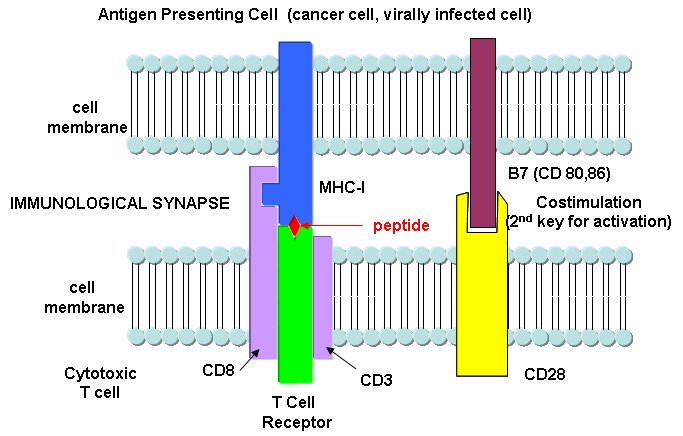
What do T cells do? What happens if a virus makes it into a cell? Antibodies can not bind to them anymore to prevent its entry. Something must be able to recognize a virally-infected cells and eliminate it. What about a cancer cell. Wouldn't it be nice if something could recognize a tumor cell as foreign and eliminate it before it divides to much and metastasizes? That something are T cells. There are many T cells in a person, and many different kinds, including T helper cells (Th) and cytotoxic lymphocytes (CTL). T cells also recognize antigens but unlike B cells, these antigens can only be proteins fragments. The membrane proteins that recognize proteins fragments are call T cell receptors. In addition, they don't recognize protein antigens in isolation. They must be bound to a protein on the surface of an "antigen" presenting cell (such as a macrophage or dendritic cell). The T cell receptor recognizes and binds simultaneously to the foreign protein fragment and to the to the self protein on the surface of the antigen-presenting cells. The self protein which binds and presents the foreign protein fragments (peptides) is called a MHC protein (Major Histocompatability Complex).
Antigen presenting cells like macrophages have MHC Class II molecules on their surface. These bind protein fragments from engulfed bacteria, for example, and present them on the surface. T cell receptors bind to the peptide:MHC II complex. Other cells in the body have MHC Class I proteins on their surface. If a cell is infected with a virus, protein fragments from the virus end up bound to the MHC Class I protein on the surface. Now a T cell can bind through its T cell receptor to the peptide:MHC Class I complex. By displaying a viral protein fragment on the surface, the immune cell can recognize a virally-infected cell without getting inside of the cell where the virus is. Sompayrac describes MHC molecules as looking like a hot dog bun. In the grove of the bun lies the peptide fragment - like the hot dog. The T cell receptor recognizes both the bun and the hot dog!
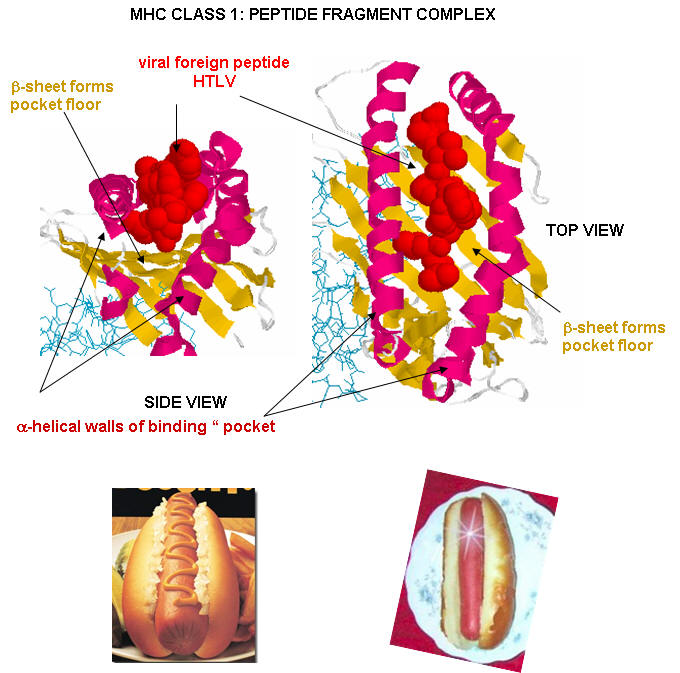
![]() Jmol
Model: The
complex between TCR A6 and human Class I MHC HLA-A2 with the modified HTLV-1
TAX (Y5(4-fluoroPhenylalanine)) peptide (Protopedia)
Jmol
Model: The
complex between TCR A6 and human Class I MHC HLA-A2 with the modified HTLV-1
TAX (Y5(4-fluoroPhenylalanine)) peptide (Protopedia)
MHC Class I proteins also present "self" peptides in the binding pocket, since self proteins also are degraded in the cell by proteasomes. However, the T cell receptor does not recognize and bind to the self-peptide fragment bound to the MHC Class 1 protein. Hence T cells do not recognize self and turn against the bodies own cells. Once and a while they do, however, and autoimmune disease like MS, rheumatoid arthritis, and lupus result.
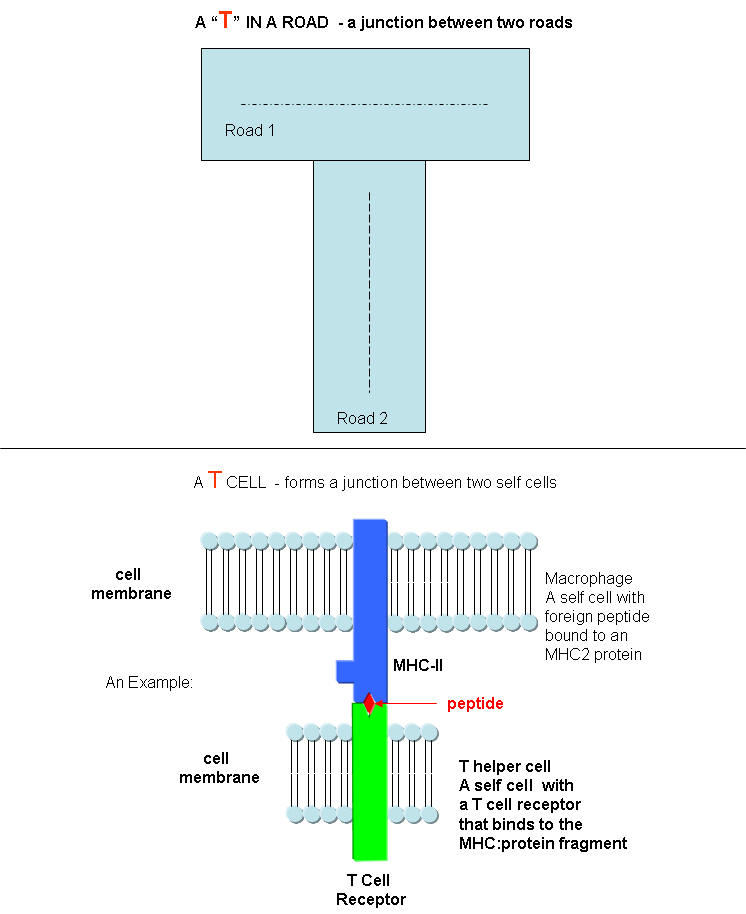
B cells and T cells must be "turned on" before the can do their thing. It is important to regulate the on switch. If the cells were to be become active without need, the cells might turn against self, which would be a big problem. It turns out that it is not enough for the T cell to bind foreign peptide:MHC complexes to get activated. They must bind another protein on the antigen-presenting cell. In the case of T helper cells. a protein on the T cell, CD28, must also bind a protein, B7, on the antigen-presenting macrophage, Hence there is one specific signal (the peptide:MHC complex binding to the T cell receptor) and a nonspecific signal (B7 binding CD28). Why are two signals needed for activation? Again Sompayrac has a great analog - that of a safety deposit box. It takes two keys (a specific key which you own and a nonspecific key (which the bank opens and which opens all the boxes) to open the box. Think of it as double security. You don't want to activate immune cells for killing unless you really need to do so. Activation of cells either in the innate or adaptive IS occurs through signal transduction processes. Either of two event participate in cell activation:
- cell:cell contact in which one cell surface protein binds a protein receptor on another
- cell activation through soluble protein signals released by immune cells. The proteins are secreted by an activated cell and bind to a receptor on another immune cells, which through signal transduction processes, gets activated. These soluble protein signals are often called cytokines. Some are called interleukins, because they travel between leukocytes - white blood cells.
Which is more important, the innate or adaptive immune system (IS)? Modern thought tilts toward the innate IS. It posts the sentinels. It gets activated when cells detect common pathogens such as bacteria, parasites, and viruses. It then activates the adaptive immune system. We'll first discuss activation of B and T cells in the adaptive immune response, and then return to the initial event, activation of the innate immune system.
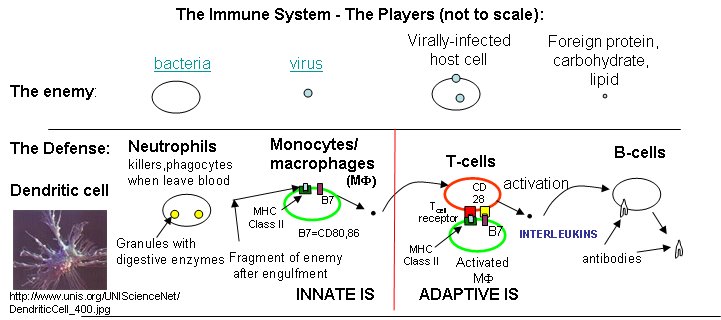
Immune cells in action
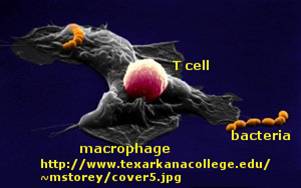
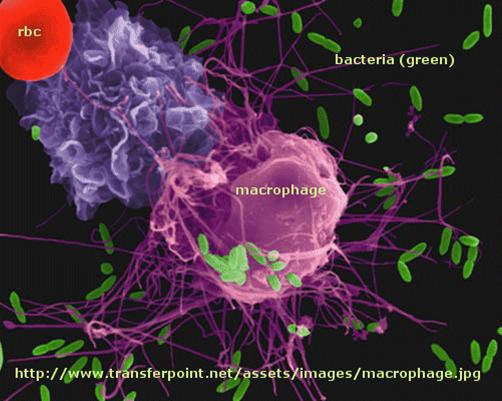
F3. T cell activation by Antigen Presenting Cells
Antigen presentation by MHC molecules on activated macrophages and dendritic cells is a bit complicated. Class I MHC proteins are found on most human cells. They present peptides (hot dogs) in the grove (buns) of the MHC protein. It turns out that not only are foreign peptides (from virus proteins for example) are presented, but also self peptides. All these peptides are generated by intracellular proteolysis of proteins - self and nonself by a structure in the cell called a proteasome. Sompayrac suggests that the MHC Class 1 proteins are like "billboards [which] advertise a sampling of all the proteins that are being made in a cell". MHC Class 1 molecules present information about proteins made in the cell. They activate CTLs which can kill the altered cell.
Cytotoxic T cells have another protein CD8, which binds to its T cell receptor. The CTL must also have a second nonspecific signaling protein (as mentioned above in the safety deposit box analogy) for cell activation.
Class II MHC proteins are found on immune cells (macrophages, etc) which present antigen to T cells, especially T helper cells. They present information about proteins found outside of the cells (such as on bacteria) but whose proteins might end up inside the antigen presenting cell like a macrophage. They activate T helper cells which, through release of signaling molecules called cytokines, activate other immune cells. B cells, before activation by antigen, express little MHCII proteins (and small amounts of B7). After activation, they express more of these proteins and can activate T helper cells.
T helper cells and CTLs differ in another way. T helper cells have another protein CD4, which binds to its T cell receptor, while CTLs have the protein CD8 bound. Both cells must also have a second nonspecific signaling protein (as mentioned above in the safety deposit box analogy) for cell activation.
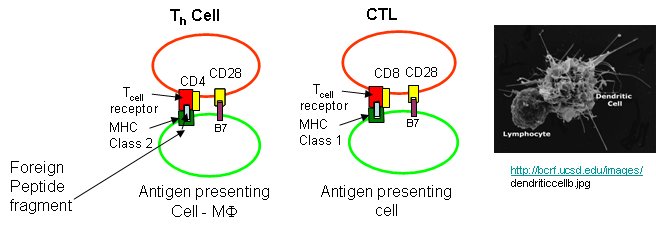
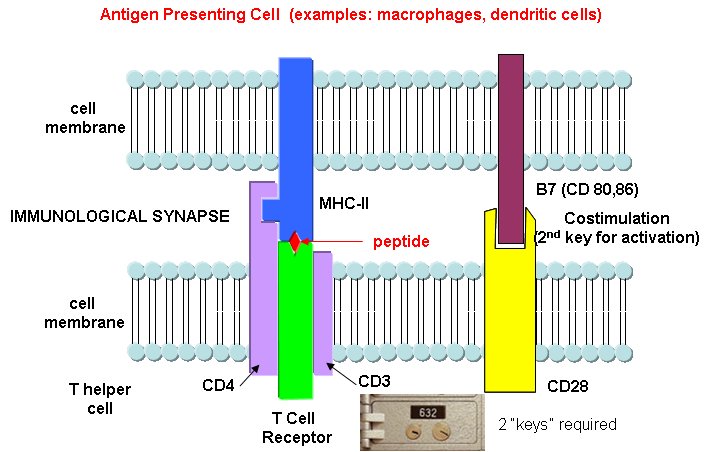
Sompayrac asks another interesting question. Why is antigen presentation by MHC proteins necessary at all? B cells don't really need presentation since they can bind antigen with membrane antibody molecules. Why do T cells need it. He gives different reasons for Class I and Class II presentations:
Class I MHC (found on most body cells): T cells need to be able to see what is going on inside the cell. When virally-infected cells bind foreign peptide fragments and present them on the surface, they can be "seen" by the appropriate T cell. It's a way to get a part of the virus, for example, to the surface. They can't hide out in the cell. T cells don't need to recognize extracellular threat since antibodies from B cells can do that. Presentation is also important since viral protein fragments that might be found outside of the cell might bind to the outer surface of a noninfected cell, that would then be targeted for killing by the immune system. That wouldn't be good. It also helps that peptide fragments are presented on the surface. This allows parts of the protein that are buried and not exposed on the surface, which would be hidden from interaction with outside antibodies, to be used in signaling infection of the cell by a virus.
MHC Class II (found on antigen-presenting cells like macrophages): In this way two different cells (the presenting cell and the T helper cell) must interact for a signal for immune system activation to be delivered to the body. Again it is a safety mechanism to prevent nonspecific activation of immune cells. Also, as in the case above, since fragments are presented, more of the foreign "protein" can contribute to the signal to activate the immune system.
F4. Cytokines and Activation of Immune Cells
There are a large number of different cytokines that are released from immune cells. Some examples and their activities are shown in the diagram below and described in the text that follows. They are
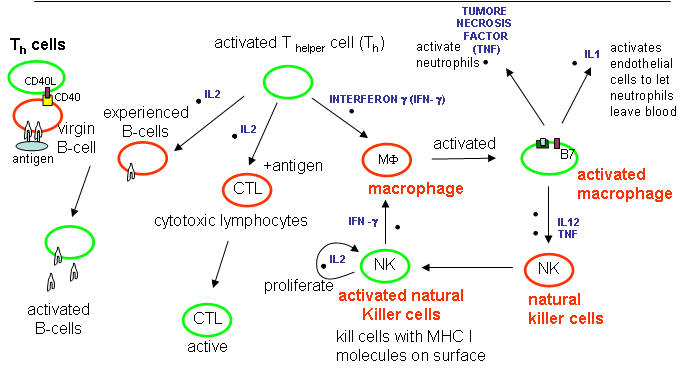
Cytokines released from cells such as macrophages in the innate IS:
Tumor Necrosis Factor: TNF - from activated macrophages and dendritic cells. Main mediator of acute inflammation. Induces fever and sleep by acting on sites in the brain. Promote scar formation (excessive wound healing). Too much can cause shock. Acts on
-
endothelial cells that line all blood vessel walls
-
facilitates the movement of immune cells from the blood into tissues through the endothelial cells.
-
activates neutrophils activates macrophages which leads to IL1 secretion kills some tumor cells.
IL-1 - has similar properties as TNF IL-12 -
-
Activates NK and CTL.
-
Leads to release of INF-g from NK and T cells.
Interferon a, b, w : Are produced by virally-infected cells.
-
cause cells to degrade mRNA which kills virally-infected cells. The enzyme involved in RNA hydrolysis is inactive until the cell becomes infected with a virus. This protects uninfected cells.
Cytokines released after antigen activation of T cells in the adaptive immune system:
IL-2 - Produced by T helper cells (CD4)
-
leads to the proliferation of NK cells and T and B-lymphocytes that have been activated by antigen exposure.
Interferon-gamma (INF-g) - is also produced by activated T cells
-
increases the activity of other T cells (CTLs, NK cells. principle way to activate macrophages.
-
induces expression of MHC-I molecules, MHC-II molecules on antigen presenting cells to promote presentation
-
IFN-g stimulates the differentiation of T4-lymphocytes into activated T helper cells.
Chemokines are chemotactic cytokines, or protein that recruit other cells towards the site of release. Some examples are listed below:
-
MIP (Macrophage inflammatory factor, CCL3,4) recruit MI, NK cells, immature dendritic cells
-
RANTES (CCL5)
-
IL-8 (recruit neutophils)
-
IP10 recruit T cells
F5. Recognition and Response in the Innate Immune System
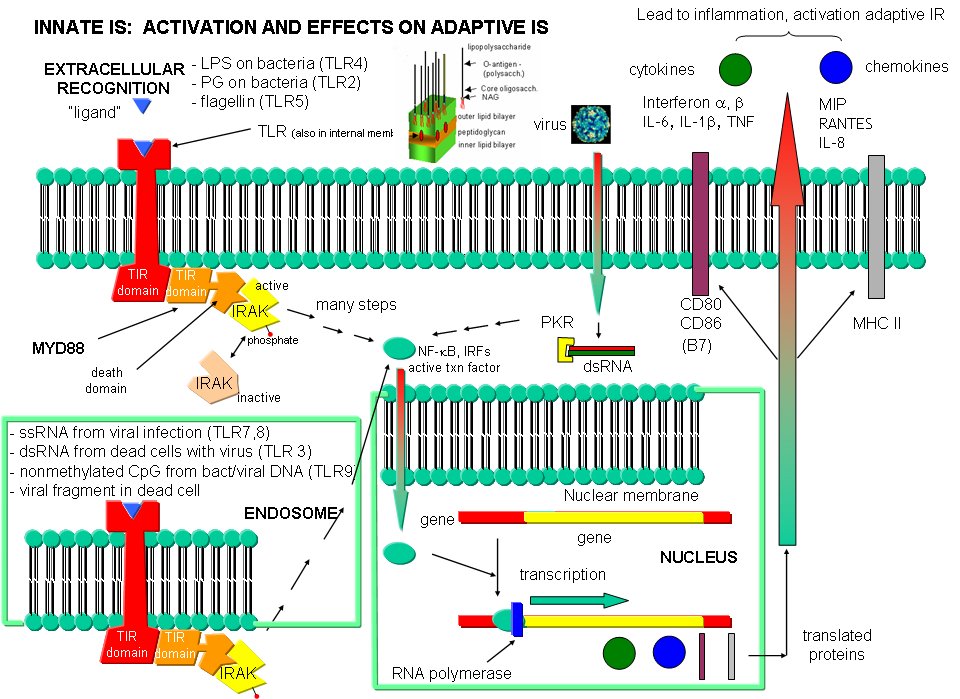
The adaptive immune response must be activated by cells of the innate immune system. These cells must recognized viruses and living cells like bacteria, fungi, and protozoans like amoebas. There are often common structural features in these different classes of cells. The cells of the innate system (dendritic cells, macrophages, eosinophils, etc) have receptors (Toll-like Receptors 1-10 or TLRs) that recognize the common pathogen associated molecular patterns (PAMPs) , which leads to binding, engulfment, signal transduction, maturation (differentiation), antigen presentation, and cytokine/chemokine release from these cells. Take for example dendritic cells, which reside in the peripheral tissues and act as sentinels. They can bind PAMPs which include:
- CHO/Lipids on bacteria surface (LPS)
- mannose (CHO found in abundance on bacteria,
- yeast dsRNA (from viruses)
- nonmethylated CpG motiffs in bacterial DNA
Bacterial and viral nucleic acids are recognized by intracellular TLRs in the cell after the they been taken up into the cells by endocytosis. Dendritic cells phagocytize microbial and host cells killed through programmed cell death (apoptosis). In the process of maturation, surface protein expression is altered, allowing the cells to leave the peripheral tissue and migrate to the lymph nodes where they activate T cells through the antigen presentation methods described above. They also control lymphocyte movement through release of chemokines. The very large figure below shows the processes involved in recognition of PAMPs by TLRs of innate immune system cells.
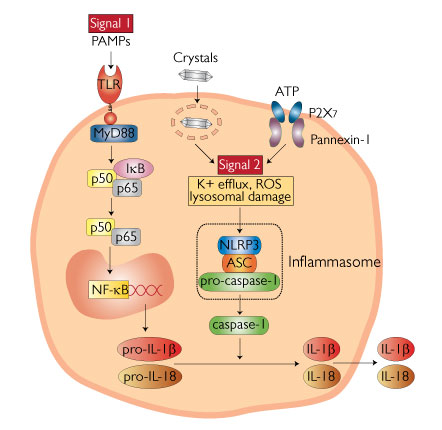
F6. Inflammasome - to be added
Figure: Inflammasome from http://www.invivogen.com/review-nlrp3-inflammasome
- The Dendritic Cell Receptor Clec9A Binds Damaged Cells via Exposed Actin Filaments Received 12 September 2011. Revised 8 February 2012. Accepted 18 March 2012. Available online 5 April 2012. Published online: April 5, 2012. Immunity http://dx.doi.org/10.1016/j.immuni.2012.03.009
- Naviaux RK, Zolkipli Z, Wang L, Nakayama T, Naviaux JC, et al. (2013) Antipurinergic Therapy Corrects the Autism-Like Features in the Poly(IC) Mouse Model. PLoS ONE 8(3): e57380. doi:10.1371/journal.pone.0057380
F7. Links and References
MHC: T cell Receptor: Peptide Complexes
![]() animation
of protein processing and display of peptides by MHC proteins on cell
surface
animation
of protein processing and display of peptides by MHC proteins on cell
surface
Preparing for the next pandemic NEJ May 5, 2005
Navigation
Return to Biochemistry Online Table of Contents
Archived version of full Chapter 5F: Immune System Recognition

Biochemistry Online by Henry Jakubowski is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.